Radiation-Induced High-Grade Spindle Cell Sarcoma Of The Sternomastoid Muscle
Explore a rare case of radiation-induced high-grade spindle cell sarcoma in the head and neck region, arising 7 years after laryngeal carcinoma treatment. Discover the challenges of diagnosis and management through combined surgery and targeted brachytherapy, leading to a successful 5-year disease-free survival. Learn about the complexities of radiation-induced sarcomas and the significance of early detection in improving prognosis.
Author:Suleman ShahReviewer:Han JuFeb 07, 20242K Shares36.5K Views
Want to know about a a rare case of radiation-induced high-grade spindle cell sarcoma? Sarcomas developing as primary malignancies of the head and neck are a rare complication after radiation therapy. This kind of sarcoma has variable clinicopathological appearances and behaviour. Radiation-induced spindle cell rhabdomyosarcoma of the sternomastoid muscle is a very rare sarcoma and has very seldom been described in the literature.
Herein, we report on the development of a rapidly growing mass over the lateral side of the neck appearing after 7 years in a patient with a historyof laryngeal carcinoma who received radiotherapy and chemotherapy. The process of diagnosis and management using combined surgery and targeted brachytherapy are discussed.
The patient experienced discomfort and oozing of the wound for up to 2 months after surgery; however, complete response with satisfactory adaptation and shrinkage of the pectoralis major pedicled muscle flap occurred after a year. The patient has been disease-free for 5 years post-operation.
Radiation-induced spindle cell rhabdomyosarcoma of the sternomastoid muscle after treatment for laryngeal carcinoma may occur a long time after radiation therapy and be clinically aggressive, radiographically distinctive and require multidisciplinary management.
Although radiotherapy is known to be an effective treatment modality in the management of malignant disease, ionizing radiation can also induce malignant tumour formation. This has been shown by studies on the sequelae of human exposure to radiation from atomic testing. The first recorded case of a radiation-induced cancer was a squamous cell carcinoma (SCC) that arose in the hand of a 33-year-old technician who had been testing roentgen tubes for 4 years.
What Is Radiation-Induced Sarcoma (RIS)?
Radiation-induced sarcoma (RIS) of the head and neck is a rare, long-term complication of treatment with radiotherapy. Although there are many case reports on RIS in the medical literature, these cases cannot provide reliable information on the outcome of pathology as there are few reported series published with only a limited number of cases.
Post-irradiation sarcoma (PIS) is another name given to RIS; the histological types include osteosarcoma, fibrosarcoma, angiosarcoma and malignant schwannoma. Spindle cell rhabdomyosarcoma (RMS) is a rare PIS first described in children in 1992 as a neoplasm composed mainly of fascicular spindle-shaped cells that show immunohistochemical and ultrastructural evidence of myogenic differentiation.
The aetiology of spindle cell RMS in adults is unknown, with different subclasses. Radiation-induced RMS is a rare variant of this type of sarcoma.
The prognosis of patients with PIS is poor in general, regardless of the type and site, with most series reporting overall five-year survival rates of 10%–30%. The prognosis appears to be related to site, reflecting the feasibility of surgical resectability. RIS of the extremities has the best prognosis, whereas that involving the vertebral column, pelvis, shoulder girdle and neck has the worst prognosis.
Although adjuvant radiotherapy and/or chemotherapy have a role in the treatment of these tumours, the extent of adjuvant radiotherapy is limited by the amount of radiation previously received, leaving surgery as the only treatment option for most of these tumours. However, using brachytherapy as an adjunctive to surgery to control residual sarcoma cells is a novel method; to the best of our knowledge, no study has been conducted using this modality.
This report describes a case of adult spindle cell RMS in a patient who had previously undergone radiotherapy, treated by combined surgery and targeted radiotherapy (brachytherapy) with a follow-up for 5 years.
Case Report
A 67-year-old Caucasian male was referred to the Head and Neck Unit, University College London Hospital, London complaining of a painless lump in the right side of the neck which had increased in size over 7 months. The mass did not cause any symptoms, including dysphagia or speech or breathing problems. Past medical history included hypertension and hypothyroidism. The patient was a non-smoker, but chronic heavy drinker (40 units/week for over 20 years).
The patient was previously diagnosed with T3N0M0 laryngeal SCC 7 years prior to his presentation to the unit. His treatment involved radiotherapy (66 Gy) and chemotherapy.
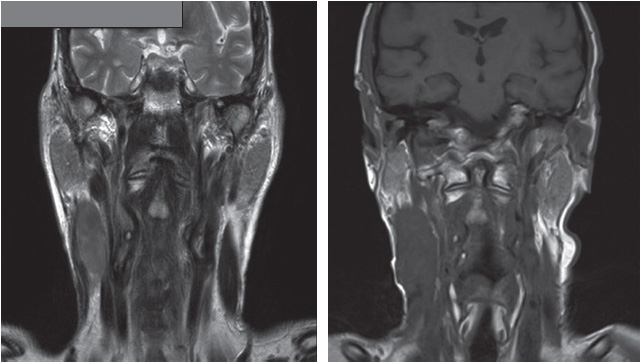
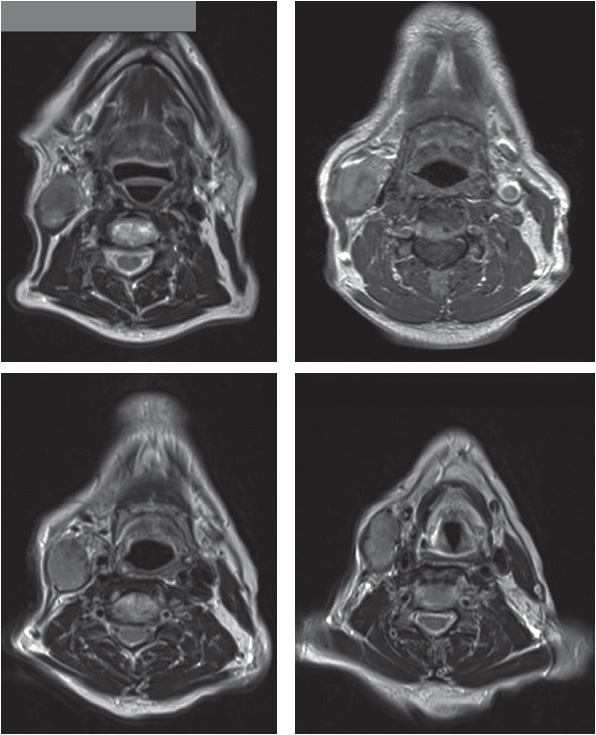
On clinical examination, the mass was 8 × 5 cm in size and infiltrating deep into the sternomastoid muscle. There were no palpable lymph nodes. Magnetic resonance imaging (MRI) of the neck reported a fixed solitary mass in the right neck (levels II and III) arising from the sternomastoid muscle, with heterogeneous high signal intensity on T2-weighted images and low signal intensity on T1-weighted images.
The patient was examined under anaesthesia that confirmed the findings; the endoscopic examination of the oropharynx and larynx was unremarkable. After an ultrasound-guided core biopsy, the diagnosis was confirmed histopathologically as radiation-induced high-grade spindle cell sarcoma.
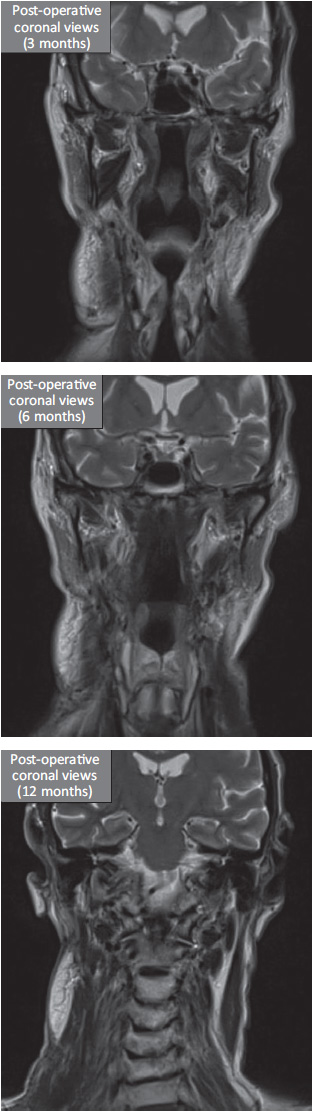
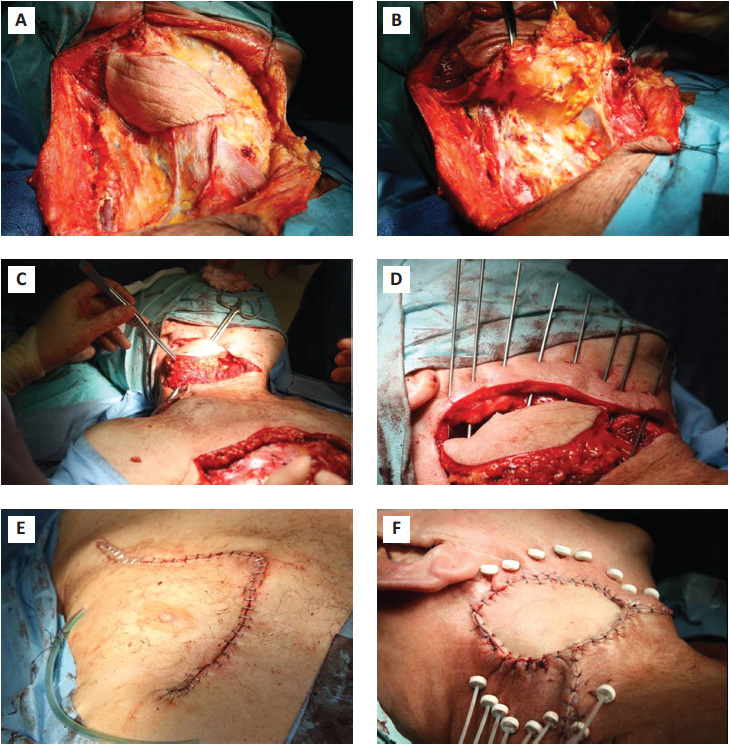
Agreement on the Multi-Discipline Meeting (MDT) was reached and the patient received single-agent doxorubicin for 3 cycles. This was followed with extended radical neck dissection with clear margins, which was successfully performed, followed by reconstruction with an ipsilateral pectoralis major pedicled muscle flap and insertion of brachytherapy catheters.
Recovery after anaesthesia was uneventful. The patient complained of discomfort in the area of surgery, with clear fluid discharge from the chest (flap donor area) for up to 2 months, which healed later with simple conservative dressings.
Macroscopically, a well-circumscribed, but not encapsulated, solid, solitary red lesion was encased in thick fibromuscular tissue. Microscopic examination revealed elongated, spindle-shaped cells with vesicular nuclei, numerous mitoses, and a pale cytoplasm, forming long fascicles. These cells were mixed with sparse polygonal or rounded rhabdomyoblasts with foci of sarcomeric differentiation. Immunohistological staining was positive for desmin, which confirmed the myogenic nature.
Radiation was given twice daily for 3 consecutive weeks. The patient had an uneventful recovery and remained disease-free at his 5-year follow-up.
Discussion
RMS is a malignant mesenchymal tumour that originates from immature cells which are destined to differentiate into striated skeletal muscle. RMS predominantly affects children, constituting >50% of all paediatric soft tissue sarcomas. It can be subdivided into three histological subtypes: embryonal, alveolar and pleomorphic.
Spindle cell RMS is a less common variant of embryonal RMS and is predominantly composed of spindle cells. The tumour is most often encountered in the paratesticular region in children, in whom it is generally associated with a better prognosis. The first two cases of spindle cell RMS in adults were described by Rubin et al. (1998).
Little is known about the aetiology of spindle cell RMS in adults. In this study, RIS developed in this patient, although radiotherapy was effective in the management of a malignant tumour of the larynx.
Determination of a cause-effect relationship between prior irradiation and radiation-induced tumour formation requires the following criteria:
- documented history of irradiation in this site
- the new malignancy must arise within the irradiated field
- the new tumour must be histologically distinct from the original primary lesion and
- the latent period between irradiation exposure and development of the new malignancy must be 5 years or longer.
Our patient fulfilled the criteria by virtue of the tumour's location, previous and new histology and latency period of >5 years. The presenting signs and symptoms of radiation-induced RMS are variable, depending on the site of initial presentation, the extent of the tumour, the presence or absence of distant metastases, and lymph node involvement. Patients generally present with a fast growing mass. Diagnosis is difficult because of induration and fibrosis of the tissue within the former field of radiation.
Generally, RISs are associated with an outcome significantly poorer than that of stage-matched soft tissue and osteogenic sarcomas that arise independently of irradiation. The 5-year disease-free survival rate for RIS is 10%–30%.
The poor prognosis of RIS in the head and neck region may be explained by the following factors:
- delay in diagnosis
- proximity of the tumour to major neurovascular structures, which may place constraints on the limits of surgical resection
- limited treatment options because of the dangers of irradiating the previously irradiated field and the relatively poor sensitivity of these tumours to chemotherapy
- RIS in the head and neck may be biologically more aggressive and (5) RIS develops from radio-resistant tumour clones and may not be responsive to further radiotherapy.
Early detection is the main hope for survival in RIS cases because there are no preventive measures to safeguard patients with cancer against RIS apart from limiting radiotherapy or using surgery as the primary treatment modality. Thus, immediate workup should be performed when any pain or swelling occurs in an irradiated field—the clinical diagnosis of RIS can be difficult due to fibrosis and induration within the irradiated field.
The management of RIS is challenging and differs from that of sarcoma not induced by radiation. Complete surgical excision appears to offer the best means of palliation and the only rational chance for long-term survival. Prognosis is related to the site and resectability; thus, RIS of the extremities has a 5-year cure rate of 30% whereas RIS of the vertebral column and pelvis is associated with a 5-year survival rate of <5%. The survival rate of RIS in the head and neck is somewhere in between these rates. In our patient, critical anatomy at the lymph node neck levels II and III did not limit us in obtaining a wide surgical margin.
Although only limited follow-up data are available, adult spindle cell RMS appears to have a more aggressive clinical course when compared with cases occurring in the paediatric population. Experience with treatment of adult spindle cell RMS is limited. Treatment options include surgery, radiation therapy and chemotherapy Patients are categorized according to their risk assessment, using a clinical group and a site-based tumour-nodes-metastasis staging system.
Radiation-Induced High-Grade Spindle Cell Sarcoma FAQs
What Is Radiation-induced High-grade Spindle Cell Sarcoma?
Radiation-induced high-grade spindle cell sarcoma is a rare type of malignancy that may develop years after radiotherapy. It predominantly occurs in the head and neck region and is characterized by aggressive growth, posing challenges in diagnosis and management.
What Are The Risk Factors For Developing Radiation-induced High-grade Spindle Cell Sarcoma?
The primary risk factor is a history of radiotherapy, particularly in the treatment of other malignancies such as laryngeal carcinoma. The latent period between radiation exposure and sarcoma development is crucial, often exceeding 5 years.
How Is Radiation-induced High-grade Spindle Cell Sarcoma Diagnosed And Treated?
Diagnosis involves a combination of clinical examination, imaging (such as MRI), and histopathological confirmation through biopsy. Treatment typically includes a multidisciplinary approach, combining surgery and targeted brachytherapy to achieve optimal results. Early detection remains critical for improving the prognosis of this rare and challenging sarcoma.
Conclusion
In conclusion, despite poor prognosis and aggressive pattern of radiation-induced high-grade spindle cell sarcoma, combining surgery with brachytherapy achieved a disease-free survival for 5 years.

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles