Extraction Of Biologically Active Compounds By Hydrodistillation Of Species Gum Resins For Anti-Cancer Therapy
A study demonstrated the probable use of gum resins for anti-cancer therapy. Frankincense essential oils from Boswellia species possess anti-cancer activity.
Author:Suleman ShahReviewer:Han JuJan 19, 2024469 Shares16.7K Views

Studies have been conducted through the years to explore the possibility of using gum resins for anti-cancer therapy, such as the traces of essential oilsthat can be obtained from them.
Essential oils produced by distillation processes from herbs and plants are traditionally confined to aromatherapy due to the abundance of low-molecular-weight, highly volatile, aromatic compounds.
Essential oils are considered to be an alternative treatment that provides supportive care to cancer patients.
Similar to the extraction process that involves the use of chemical solvents, essential oils produced from distillation procedures contain high-molecular-weight and biologically active compounds from plant materials.
The aim of this review is to determine whether the use of biologically active compounds extracted from the Boswelliaspecies gum resins is beneficial in anti-cancer therapy.
Preliminary Discussion
Aromatic gum resins obtained from trees of the genus Boswellia(family Burseraceae), also known as frankincense, have long been used in Ayurvedic and traditional Chinese medicines to treat a variety of healthproblems.
In addition to their anti-inflammatory activity, frankincense extracts have been shown to suppress tumor development and induce tumor apoptosis in animal models.
In a human clinical study, a gum resin extract prepared from Boswellia serratawas found to reduce cerebral edema, with anti-cancer activity in patients irradiated for brain tumors.
Boswellic acids have been emphasized as the major component responsible for frankincense extract-mediated anti-tumor activity.
In both in vitro and in vivo models, purified boswellic acids, particularly acetyl-11-keto-β-boswellic acid (AKBA), exhibit potent cytotoxic activities against:
- cultured human neuroblastoma cell lines
- colon cancer cells
- fibrosarcoma cells
- hepatoma cells
- leukemia cells
- melanoma cells
- meningioma cells
- pancreatic cancer cells
- prostate cancer cells
These studies demonstrate that gum resins of Boswelliaspecies contain active ingredients that have potent anti-cancer activity.
Essential oils are extracted by distillation, a process used to extract biologically active compounds from plant materials.
In addition to classical uses of essential oils in aromatherapy, we have studied the anti-tumor activities of frankincense essential oil extracted by hydrodistillation of gum resins from Boswelliaspecies.
We have demonstrated that frankincense essential oil is highly effective in suppressing the proliferation and inducing the cytotoxicity of various human cancer cell lines in cultures and in an animal model; however, such anti-cancer effects cannot be entirely attributed to boswellic acids.
This review is based on the extraction of biologically active compounds from gum resins of the Boswelliaspecies for anti-cancer therapy.
Main Discussion
Boswellia Sacra Gum Resins And Hydrodistillation
Frankincense resins are harvested from deep incisions made into the tree trunk of Boswellia species.
This wounding process causes the tree to “bleed” a milky white substance that seals and heals the wound to prevent infection.
To certify the harvest location, species and classification, B. sacragum resins were obtained directly from local collectors in the Dhofar Mountain region surrounding Salalah, Sultanate of Oman.
To prepare frankincense essential oil, the B. sacragum resin was processed in a custom-made hydro-distiller in Salalah, Oman.
The typical yield of frankincense essential oil by hydrodistillation is 10% (w/w) of gum resins within a range of 8% to 13%.
Chemical Components
Chemical composition and optical rotation of frankincense essential oils from Boswelliaspecies are determined using chiral gas chromatography-mass spectrometry (GC-MS) and polarimetry, respectively.
Chemical constituents of frankincense essential oils depend on geographical locations of trees, transportation, storage and processing procedures.
We have distinguished the chemical properties (P < 0.0001) of the essential oils hydrodistilled from the gum resins of B. sacraand Boswellia carterii.
B. sacraessential oil possesses a positive optical rotation of polarized light (+30.1° with standard deviation of 5.4° for n = 88), while B. carteriiessential oil is characterized with a negative optical rotation (-13.3° with standard deviation of 4.9° for n = 39).
In addition, chiral GC-MS of the monoterpenes (especially the most abundant α-pinene: 79.0% in B. sacraand 48.2% in B. carterii) with a chiral centre demonstrated the reason for the differing optical rotations.
The enantiomeric ratio of the (+)/(-) enantiomers of α-pinene was 8.24 for B. sacraessential oil versus 0.68 for B. carteriiessential oil.
Four other chiral monoterpenes had a greater abundance of the (+) enantiomer in B. sacraessential oil with (+)/(-) enantiomeric ratios ranging from 2.77 to 7.00, whereas the (-) enantiomer was more abundant in B. carteriiessential oil with (+)/(-) enantiomeric ratios ranging from 0.04 to 0.85.
We have found that a quick measurement of the optical rotation by polarimetry can provide additional evidence of the local geographic source of the gum resins.
For instance, B. sacraresins harvested from trees in the westerly Mughsayl area of the Dhofar Mountains yield essential oil distinctive of the easterly Hasik area gum resins.
Mughsayl (black) frankincense essential oil has a significantly higher percentage (P < 0.0001) of α-pinene (79.0% vs. 48.2%) noted by a similarly higher optical rotation of +30.2° versus +19.3°.
Conversely, Hasik area frankincense essential oil contains higher quantities of myrcene (11.4% vs. 1.1%) and limonene (13.6% vs. 3.7%).
The higher limonene content of Hasik area gum resins provides a “sweet, orange” aroma that makes it more valuable to resin-burning consumers.
These results have been confirmed by a recent report that α-pinene is the principal component and chemotaxonomical marker to identify the botanical and geographic source of B. sacraresins within the Dhofar Mountain range of Oman.
Hydrodistillation procedures determine the chemical constituents of the frankincense essential oil.
For example, the abundance of high-molecular-weight compounds is positively associated with distillation time and temperature.
Longer duration and higher temperature distillations produce greater amounts of high-molecular-weight compounds, such as sesquiterpenes and boswellic acids.
There are time-dependent and temperature-dependent chemical compositions in frankincense essential oils.
Based on GC–MS profiling, frankincense essential oil obtained at 100 °C for 24 hours consists of more than 300 different identifiable compounds ranging in mass from 72 to 468 amu (atomic mass unit).
Molecular Mechanism Of Frankincense Essential Oil-Induced Cancer Cell Apoptosis
We have demonstrated that frankincense essential oils prepared from hydrodistillation of B. carteriiand B. sacra gum resins at 100 °C for 24 hours possess potent growth suppression activity in cultured:
- breast cancer cells
- colon cancer cells
- human bladder cancer cells
- pancreatic cancer cells
The growth-suppressing activity of frankincense essential oils results from a combination of anti-proliferative and pro-apoptotic activities.
In addition, frankincense essential oil overcomes multicellular resistance and suppresses invasive phenotypes of human cancer cells.
More importantly, human tumor cell lines are more sensitive to frankincense essential oil-induced growth arrest and apoptosis as compared with their normal counterparts, and similar results have been observed in cell lines derived from the:
- bladder
- breast
- colon
This is an important and unique characteristic of frankincense essential oil as compared with other essential oils, including lavender and sandalwood.
Consistent with results from cell viability assays, several gene products identified as frankincense essential oil-responsive genes are associated with suppression of cell proliferation and arrest of cell cycle progression.
Frankincense essential oil up-regulates the expression of anti-proliferative genes, including:
| CLK1 | IL8 |
| CDKN1A | KLF4 |
| DLG1 | NEDD9 |
| IL1A | SNFILK |
| IL6 | SSTR1 |
It does the same thing on cell cycle arrest genes, including DDIT3, IL8, and CDKNIA in human bladder cancer cells.
We have also demonstrated that frankincense essential oil suppresses the expression of cyclin D1 and cdk4 proteins in human cancer cell lines, and may result in blocking G1/S transition in cell cycle progression in these cells.
Frankincense essential oil also up-regulates genes in human bladder cancer cells as well as a number of genes that are responsible for apoptosis, which include:
| CDKN1A | IL6 |
| DEDD2 | NUDT2 |
| IER3 | SGK |
| GAD45B | TNFAIP3 |
In addition, expression of a cell survival gene AXL is suppressed by frankincense essential oil.
Consistent with results from boswellic acid-treated HT29 and HepG2 cells, frankincense essential oil-induced apoptosis is caspase-dependent based on the cleavages and activation of the following in various human cancer cell lines:
- caspase-3
- caspase-8
- caspase-9
- poly(ADP-ribose) polymerase
Frankincense essential oil activates multiple signaling pathways in cultured human cancer cell lines.
We have shown that frankincense essential oil increases the levels of Akt phosphorylation at Ser(473) and enhances Erk1/2 activation in human breast and pancreatic cancer cells.
These results are consistent with reports that boswellic acids and AKBA activate the PI3K/Akt pathway in colon cancer HT29, HCT-116, SW480, and LS174T cells and Erk1/2 in human polymorphonuclear leukocytes and platelets.
Activation of Akt and Erk1/2 pathways in cancer cells by anti-cancer compounds with pro-apoptotic activity have also been reported.
Significances of frankincense essential oil-modulated PI3K/Akt and Erk1/2 pathway activation in inducing tumor cell growth arrest and apoptosis require further studies.
Frankincense essential oil-modulated anti-tumor activity is also observed in vivo.
Subcutaneous administration of frankincense essential oil significantly suppresses tumor growth and progression in a heterotopic xenograft human pancreatic cancer mouse model.
In agreement with frankincense essential oil-induced anti-proliferative and pro-apoptotic activities in cultured cancer cells, administration of frankincense essential oil suppresses the number of phospho-histone H3 (Ser10)-positive proliferative cells.
It also increases the number of terminal deoxynucleotidyl transferase dUTP nick-end labeling-positive apoptotic tumor cells as compared with the control group.
Identification Of Possible Anti-Cancer Components In Frankincense Essential Oil
Frankincense essential oil obtained from hydrodistillation of B. sacra gum resins has been studied to analyze the relationship between chemical composition and anti-cancer activity.
We have reported that the abundance of high-molecular-weight compounds is positively correlated with frankincense essential oil-induced cytotoxicity in cultured human cancer cell lines.
For example, frankincense essential oil prepared from distillation at higher temperature contains higher amounts of heavy molecular weight compounds and possesses more potent anti-cancer activity.
In addition, differential fractionations of frankincense essential oil produce reduced levels of low-molecular-weight compounds, and are significantly more potent than unfractionated essential oil in suppressing tumor cell viability.
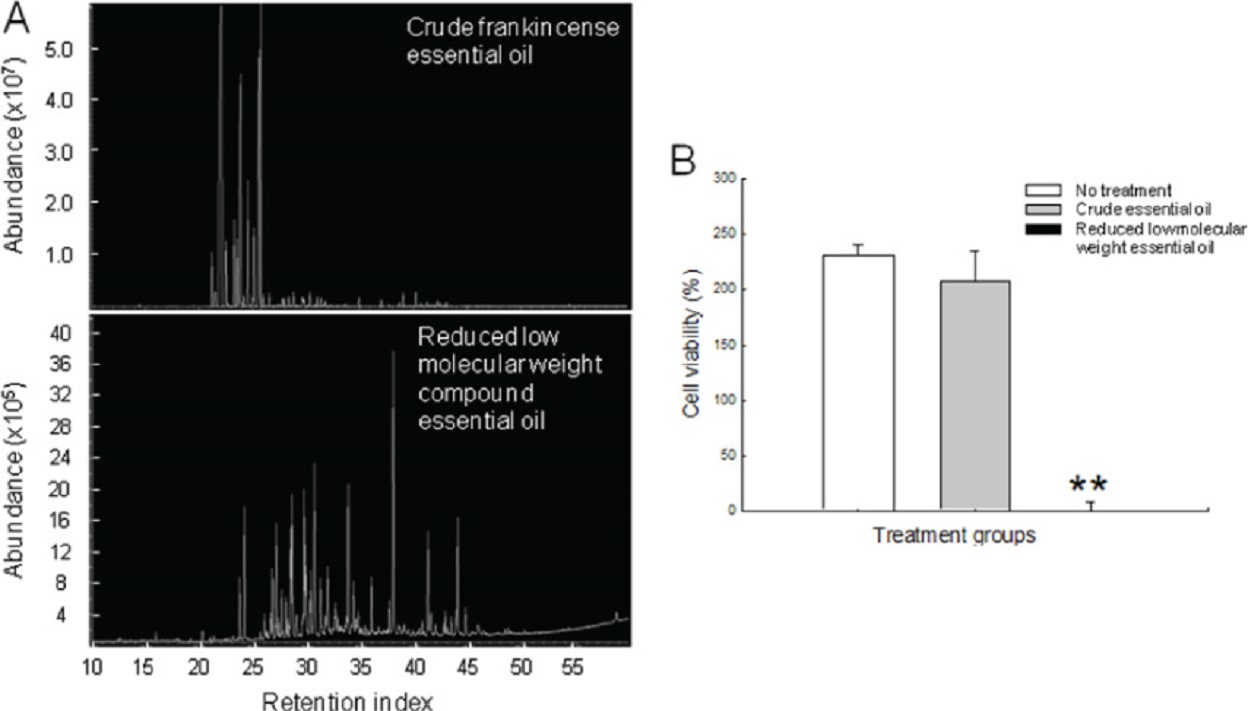
These results suggest that high-molecular-weight compounds and/or ratios of these compounds play important roles in frankincense essential oil-mediated anti-tumor activity.
Although boswellic acids, especially AKBA, have been enriched or purified from Boswellia species gum resins for studying their anti-cancer property, our results suggest that compounds other than boswellic acids might be equally or more important in frankincense essential oil-modulated anti-cancer activity.
For example, contents of boswellic acids in frankincense essential oils are not proportionally related to essential oil-induced tumor cell death.
We also observed that frankincense essential oil enriched with high-molecular-weight compounds but with lower boswellic acid contents is significantly more potent in suppressing the viability of cultured human cancer cells.
Additionally, frankincense hydrosol, the aqueous distillate of hydrodistilled Boswelliaspecies gum resins, contains up to 15.5% boswellic acids but does not have detectable cytotoxicity against tumor cells.
Another compound, tirucallic acid, purified from B. carteriigum resins, has been found to induce human prostate cancer cell death.
With a complex chemical constituent in frankincense essential oil, many components may work synergistically to provide a potent anti-cancer activity.
Although it might not be easy to isolate a group of compounds working synergistically, it is important to identify a signature compound(s) that assures consistent biological and anti-cancer activities.
Essential oils for cancer patients have been primarily emphasized as a supportive care for general well-being.
Uses of essential oils are generally limited to inhalation or topical application in the United Kingdom and the United States; however, essential oils can be administered orally or internally in France and Germany.
Since the distillation process is intended to extract biologically active compounds from plant materials without the aid of chemical solvents, essential oils shall contain chemical constituents similar to those extracted from chemical solvents but with higher portions of low-molecular-weight compounds.
Thus, essential oils shall be suitable for ingestion as long as signature compounds can be defined, and safety profiles can be established.
Based on a recent report, topical applications of frankincense essential oil distilled from B. carteriigum resins have been shown to be effective in the treatment of malignant melanoma in horses.
A similar approach can be applied to patients with bladder cancer, if appropriate doses of frankincense essential oil can be administered into the bladder through instillation.
Frankincense gum resins are used as a component in anti-cancer drugs in traditional Chinese medicine.
A chemical extract of B. serratagum resins has been investigated as an anti-cancer agent in patients irradiated for brain tumors, with no severe adverse effects being observed.
In an ongoing phase II clinical trial, a chemical extract of Boswelliaspecies gum resins is studied as an adjuvant agent in patients with high-grade gliomas.
Frankincense essential oil obtained from B. sacragum resins has been further fractionated and administered to patients suffering from various types and stages of cancers in Arabic nations for oral ingestion.
The fractionated frankincense essential oils have lower levels of low-molecular-weight compounds (proprietary information), and have been shown to have reduced adverse symptoms, including:
- diarrhea
- light-headedness
- irritated stomach
Patients responded positively after receiving a defined volume with specific chemical components of frankincense essential oil for a specific period of period.
To prepare frankincense essential oil for anti-cancer therapy, standardized procedures to harvest, store and process Boswelliaspecies gum resins have to be established to obtain consistent results.
In addition, before the active ingredient(s) in frankincense essential oil is identified, standardization of the product using GC-MS and high-performance liquid chromatography is our best approach to assure quality and efficacy.
Although no serious safety and toxicity issues have been raised in animal models and patients receiving oral administration of frankincense extracts, the safe dose of essential oil needs to be defined for cancer therapy.
Moreover, pharmacokinetics and pharmacodynamic properties of the essential oil need to be determined.
Course to cancer recovery using frankincense essential oil may be very different from our current understanding of tumor regression using:
- chemotherapy
- radiation therapy
- targeted therapy
The frankincense essential oil may activate the hosts’ inflammatory responses to suppress tumor progression by its anti-inflammatory activity.
Thus, additional clinical studies are required to understand the mechanism of frankincense essential oil-suppressed tumor progression in human subjects.
Conclusion
Frankincense essential oil obtained from the hydrodistillation of Boswelliaspecies gum resins possesses anti-cancer properties by activating:
- multiple signaling pathways
- cell cycle regulators
- caspase-dependent apoptosis
Safety and efficacy, pharmacokinetics and pharmacodynamics, as well as tumor and patient responses of systemic frankincense essential oil administration need to be studied.
Frankincense essential oil can be a novel and alternative therapeutic agent to suppress cancer progression and metastasis with minimal adverse effects when an appropriate dose is defined.
Results from preliminary clinical observations suggest the potential effectiveness of gum resins for anti-cancer therapy.

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles