Nephropathies In HIV-Infected Patients - An Overview
Through the years, the incidence of nephropathies in HIV-infected patients have risen. Kidney disease has emerged as a significant cause of morbidity and mortality for them.
Author:Suleman ShahReviewer:Han JuJan 19, 20242.8K Shares52.5K Views
The aim of this review was to discuss nephropathies in HIV-infected patients.
Over the last two decades, the number of individuals infected with human immunodeficiency virus (HIV) has markedly increased.
Since the introduction of the highly active antiretroviral therapy(HAART) at the end of 1995, the number of deaths caused by HIV infection or by an acquired immunodeficiency syndrome (AIDS)-defining disease has dramatically decreased.
HAART has increased survival and therefore extended the mean age of our population.
However, this prolongation has been accompanied by the emergence of:
- acute and chronic kidney disease (CKD)
- subsequent end-stage renal disease (ESRD)
CKD and ESRD have then become major causes of morbidity and mortality in HIV-infected patients.
Now, nephrologists are faced with several challenges regarding kidney disease in HIV-infected populations, including:
- identifying early signs of kidney disease
- working in collaboration with HIV experts to provide the best treatment to patients with renal disease
Renal disease is becoming an increasingly prevalent entity in HIV-infected patients and occurs at all stages of HIV infection.
Renal pathology in HIV patients can be caused by a variety of mechanisms creating a broad spectrum of clinical disease.
HIV-related renal impairment can present as acute or chronic kidney disease.
It can be caused directly or indirectly by HIV and/or by drug-related effects that are directly nephrotoxic or lead to changes in renal function by inducing metabolic vasculopathy and renal damage.
Acute Renal Failure In HIV-Infected Patients
RIFLE and AKIN (the acronyms for diagnosis and classification of acute renal dysfunction) criteria are frequently used.
They aim to standardize the definition of acute kidney injury (AKI) by stratifying patients based on:
- changes in serum creatinine levels from baseline; and/or
- an abrupt decrease in urine output
Tubular dysfunction is defined as an abnormal presence of markers in urine, for example:
- hyperaminoaciduria
- euglycemic glucosuria
- beta2-microglobulinuria
- hyperphosphaturia
AKI is a common finding in HIV-infected patients and has been associated with advanced stages of HIV infection, for example:
- CD4 cell count of < 200 cells per cubic millimeter
- HIV RNA (ribonucleic acid) level of > 10,000 copies per milliliter
It is also associated with:
- prior renal impairment
- hepatitis C virus (HCV) co-infection
- liver disease
- a historyof HAART
HIV-infected patients are also at increased risk for AKI development related to:
- volume depletion
- sepsis
- radiocontrast
It is also related to the administration of nephrotoxic medications used in the treatment of opportunistic infections, such as:
- antibiotics (aminoglycosides)
- antifungals (amphotericin B)
- antivirals (acyclovir, ganciclovir)
- anti-tuberculosis drugs
- pentamidine
- anti-inflammatory drugs
In a study published in 2006 by the journal Clinical Infectious Diseases, the authors, with Dr. Jorg Roling as lead author, evaluated the incidence and etiology in a prospective analysis of 754 HIV-infected patients.
Dr. Roling and his co-authors reported an incidence of 5.9 cases of AKI per 100 patient-years.
In the HAART era, the reported incidence of AKI in hospitalized HIV-infected patients ranged from 6% to 20%, and AKI was associated with increased in-hospital mortality.
Acute interstitial nephritis (AIN) can occur as a result of HIV infection of the kidney itself, as in 28% of autopsy findings in HIV-infected patients.
Interruption of the potential liable agent is the pivot of therapy.
These complications can be prevented or minimized with wide fluid intake. In severe cases, immunosuppressive therapy has been employed.
Electrolyte disturbances of the following are common:
- hypo-hypernatremia
- hypophosphatemia
- hypocalcemia
- hypomagnesemia
Hyponatremia is often observed in HIV-infected patients with gastroenteritis.
The syndrome of inappropriate antidiuretic hormone secretion (SIADH) is usually due to intracranial and respiratory infections, such as:
- pulmonary tuberculosis (TB)
- pneumocystis pneumonia
- toxoplasmosis
Hypokalemia is also often found. This case of low potassium level is due to:
- gastrointestinal losses
- renal tubular loss
- severe malnutrition
Toxicity from HAART such as tenofovir can cause:
- Fanconi syndrome
- nephrogenic diabetesinsipidus
Therefore, the dosing of nephrotoxic drugs should be adjusted to the estimated glomerular filtration rate (eGFR) in patients with acute or chronic kidney damage.
There is a higher prevalence of urinary tract infections (UTIs) and seem to be more due to malnutrition than from immunosuppression due to HIV infection.
Pulmonary and disseminated tuberculosis should be included in the differential diagnosis of acute or chronic renal failure. The following viral infections should also be included:
- cytomegalovirus (CMV)
- hepatitis B
- hepatitis C
It has been shown that patients who survive AKI have a greater rate of long-term mortality.
In a study published in 2010 by the journal Kidney International, the authors, with Andy I. Choi as lead author, evaluated the long-term consequences of AKI in 17,325 HIV-infected patients during their first hospitalization.
Over a mean follow-up period of 5.7 years, they found that AKI was associated with:
a. increased mortality
b. long-term risk of:
- heart failure
- cardiovascular disease
- end-stage renal disease
Chronic Renal Disease In HIV-Infected Patients
The prevalence of chronic kidney disease (CKD) in the various stages of HIV infection is difficult to assess.
In a study of 2,038 female HIV-infected patients and published in 2004 by the journal Clinical Infectious Diseases, the authors, with Lynda Anne Szczech as lead author, proteinuria and elevated creatinine level have been found in 7.2% to 32% of HIV-seropositive patients.
Autopsy studies yield a prevalence of up to 43% of pathological changes on histological examination.
The cause of CKD in HIV-infected patients can be difficult to assess on clinical grounds alone and can most often only be determined by renal biopsy.
CKD is defined by KDIGO (Kidney Disease Improving Global Outcomes) and is divided into groups based on confirmed eGFR levels (≥3 months).
CKD can be caused by multiple pathophysiological mechanisms.
HIV itself seems to directly mediate the development of HIV-associated nephropathy (HIVAN) and thrombotic thrombocytopenic purpura.
One of the main causes of CKD is HIVAN, a clinicopathologic entity characterized by:
- severe proteinuria
- renal failure
- rapid progression to ESRD
- frequently enlarged kidneys visible on renal ultrasound
HIVAN was initially described by the authors of a case report published in 1984 by The New England Journal of Medicine, with Dr. T. K. Sreepada Rao as lead author.
Dr. Rao, a nephrologist, and his co-authors reported a pattern of focal segmental glomerulosclerosis, often of the collapsing variant, and microcystic tubulointerstitial disease in HIV-seropositive patients in New York City.
It was directly linked to infection of epithelial cells by HIV, and was recently found to be related to polymorphisms in the APOL1 gene.
Indeed, almost all patients developing HIVAN are of African origin.
Renal biopsy is the only means of establishing the diagnosis of HIVAN.
Without adequate treatment, the prognosis of HIVAN is poor.
Risk factors for the development of HIVAN include a:
- CD4 cell count < 200 cells per cubic millimeter
- high viral burden
Pharmacologic agents used for the treatment of HIVAN include:
- steroids
- highly active antiretroviral therapy (HAART)
- angiotensin-converting enzyme (ACE) inhibitors
Recently, cyclosporine has been used as another option in children, but clinical experience with it is limited.
The introduction of HAART was shown to be associated with a reduction in HIVAN incidence.
Thrombotic microangiopathy (TMA), hemolytic uremic syndrome (HUS), and thrombotic thrombocytopenic purpura (TTP) present a spectrum of diseases characterized by:
- hemolytic anemia
- thrombocytopenia
- renal insufficiency
They are also characterized by clinical features, such:
- fever
- neurological manifestations
Several reports have linked TMA to HIV infection, suggesting that HIV proteins may mediate endothelial dysfunction, leading to platelet deposition in the microvasculature.
Therapeutic options consist of plasma infusion and plasmapheresis, which have had variable success.
Although general treatment recommendations are lacking, other attempted therapies include:
- glucocorticoids
- immunoglobulin infusions
- antiplatelet drugs
- vincristine
- splenectomy
Other pathophysiological pathways comprise indirect viral effects, such as various forms of deposition of HIV immune complex kidney (HIVICK) disease.
The prevalence of HIVICK is highly variable in the different studies.
A study of 60 biopsies found that some form of HIVICK was present in 37% of biopsy specimens.
HIVICK may present as post-infectious glomerulonephritis and includes:
- membranous nephropathy
- IgA nephropathy
- lupus-like glomerulonephritis
- fibrillary glomerulonephritis
- immunotactoid glomerulopathy
- membranoproliferative glomerulonephritis
Cryoglobulinemic membranoproliferative glomerulonephritis (MPGN) is the most characteristic renal disease associated with HCV infection.
Indeed, HCV co-infection in HIV patients is a very common problem, affecting approximately 30% of HIV-infected patients.
Patients with HIV IC seem to benefit from treatment with:
- ACEi
- glucocorticoids
- antiretrovirals
CKD is a serious complication of long-term intravenous drug use (IVDU).
Renal disease-related IVDU has been reported since the 1970s, mostly in the context of heroin-associated nephropathy, characterized by:
- nephritic syndrome
- rapid progression
Two recent studies from Europe observed changing patterns of renal disease in patients with IVDU, with concomitant chronic HIV, HBV, and HCV infections, reporting an increased prevalence of renal AA-amyloidosis.
Renal AA-amyloidosis is a complication of chronic and/or recurrent inflammatory disease. The typical clinical manifestations of renal AA-amyloidosis are:
- severe proteinuria
- nephrotic syndrome
- renal insufficiency
Successful treatment of the underlying inflammation, by immunosuppressants for autoimmune diseases or by antimicrobials for chronic infections, can lead to stabilization of or even improvements in renal function.
Renal Adverse Effects Of HAART
The use of HAART has changed the natural history and spectrum of kidney disease in HIV-positive patients.
When it comes to HIV-related complications, HAART is effective in controlling:
- viral replication
- restoring immune function
- decreasing their occurrence
However, it may be associated with nephrotoxicity or with increased rates of:
- dyslipidemia
- hypertension
- diabetes
HAART is also associated with an increase in secondary renal damage, such as:
- hypertensive nephrosclerosis
- diabetic glomerulopathy
- vascular complications
Antiretroviral therapy has been associated with impaired glucose tolerance.
A study of 17,852 HIV-seropositive patients evaluated a prevalence of diabetes of 2.5% that was significantly associated with antiretroviral treatment.
Another study that observed a cohort of 5,578 patients from 1984-2003 revealed an incidence of hypertension in 7.3% of HIV-seropositive patients.
Renal damage caused by HAART can result in a variety of toxic drug effects presenting as:
- acute renal failure
- tubular necrosis
- kidney stones
- chronic kidney disease (CKD)
HAART-associated nephrotoxicity has been described in many case reports and systematic observational studies.
Most studies, however, conclude that the occurrence is relatively low but warrant clinical attention due to the potentially serious complications.
Choi et al. found that HAART-related nephrotoxicity accounted for 14% of all AKI cases among HIV-positive individuals.
Another study, including 60 kidney biopsies performed during AKI, Roling et al. found 5% were HAART-related.
For tenofovir, Gilead has reported a 0.5% incidence of serious adverse renal events.
There are several direct and indirect ways in which HAART can induce damage to renal structures..
There is an ongoing and unresolved discussion whether HAART-induced nephrotoxicity is reversible.
Several studies demonstrate that only some patients reached pre-exposure levels, whereas several safety studies indicate a faster and more complete resolution after discontinuation.
The contribution of HIV as well as HAART to bone disease remains unclear, which may include:
- loss of bone mineral content
- aseptic necrosis of the femoral head
HIV patients are at an increased risk of osteoporosis, which is four times higher than the general population.
According to the information provided by Italian Cohort Naive Antiretrovirals (ICONA), more than54% of patients with HIV infection lack vitamin D.
The virus has a direct responsibility in the pathogenesis of osteoporosis by acting on:
- osteoblasts
- osteoclasts
HAART also promotes the toxic action of the HIV virus on bone, as demonstrated by:
- a study by Todd T. Brown and Roula B. Qaqish and published in 2006 by the journal AIDS
- the SMART (Strategies for Management of Antiretroviral Therapy) study
These drugs act on the mitochondrial DNA, resulting in an increased production of lactic acid.
Chronic metabolic acidosis causes an increase in osteoclastic activity and a reduction in osteoblastic activity.
Acid-base disturbances are common in HIV-infected patients and are mainly due to drugs and sepsis.
Lactic acidosis may possibly be due to drug-induced mitochondrial dysfunction reported with:
- zidovudine
- stavudine
- lamivudine
- didanosine
It could also be present in a mild form in 5% to 25% of patients.
Non-anion gap metabolic acidosis can result from:
- intestinal loss of bicarbonate due to diarrhea
- renal losses due to drug toxicity (most commonly amphotericin B and tenofovir)
Types Of Studies
All randomized controlled trials (RCTs) and quasi-RCTs evaluate the current knowledge on:
- acute and chronic HIV-associated renal disease
- metabolic alterations and related nephropathies
- toxic drug effects of HAART
Electronic Searches
We checked the reference lists of nephrology textbooks, review articles, and relevant studies as well as these sources:
- Cochrane Renal Group’s Specialized Register
- AEGIS (AIDS Education Global Information System) database
- ClinicalTrial.gov
- WHO International Clinical Trials Registry Platform (ICTRP)
- the U.S. National Library of Medicine’s (NLM) MEDLIN
Results
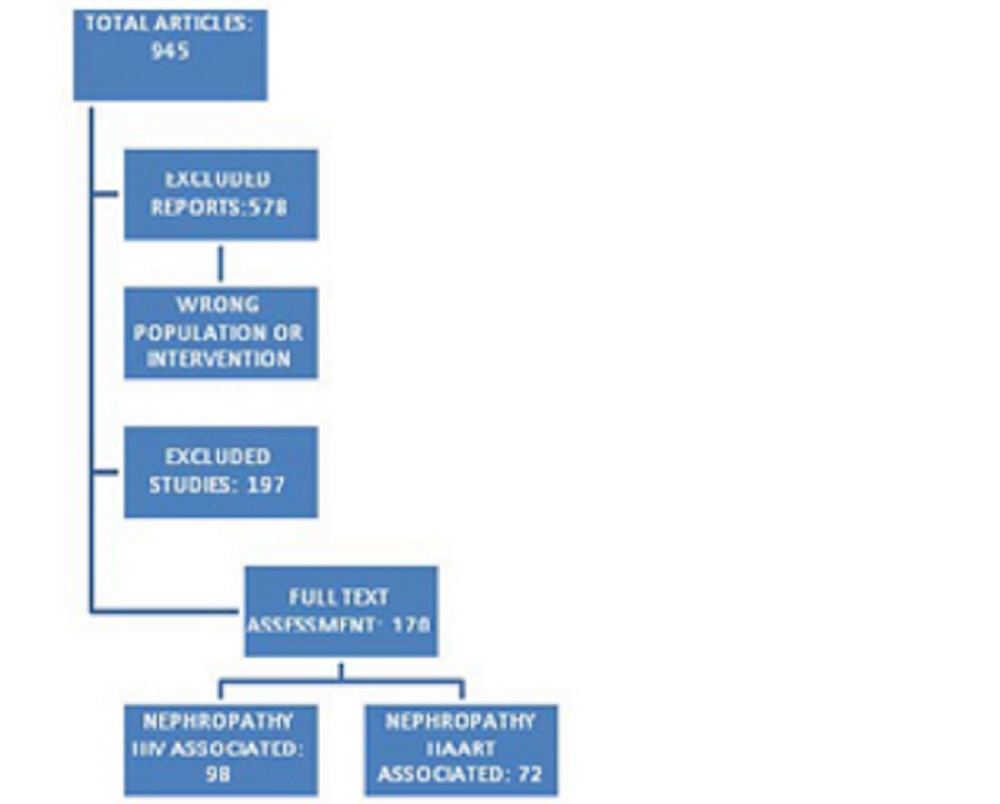
We analyzed 945 studies, out of which 578 were excluded for wrong population or intervention and 197 deemed unfit.
After quality appraisal, 170 met the inclusion criteria for the review.
The studies included in the review were grouped into two areas:
- nephropathy HIV associated
- nephropathy HAART associated
Discussion
The incidence and spectrum of kidney disease in HIV-infected patients has changed considerably over the years.
Identification of the cause of renal damage may be crucial in directing specific treatments.
Renal biopsy should be considered in all HIV-infected patients with proteinuria, whatever their renal function, CD4+ count, or viral load.
Through the use of ultrasound-guided renal biopsy and automatic biopsy devices, percutaneous renal biopsy has become safe.
Transjugular renal biopsy may represent a relatively safe and reliable alternative to conventional percutaneous biopsy in patients with risk factors for bleeding, such as thrombocytopenia.
Periodic monitoring of albuminuria, tubular parameters, and eGFR may be useful in the early diagnosis of patients at risk for acute or chronic kidney disease.
Estimates of kidney function have not been thoroughly validated in HIV-infected individuals and may be insensitive to early decrements in kidney function.
The gold standards for determination of renal function are expensive and inconvenient.
Although the current Infectious Diseases Society of America (IDSA) guidelines recommend using the Modification of Diet in Renal Disease (MDRD) equation to estimate kidney function in HIV-infected individuals, Chronic Kidney Disease-Epidemiology Collaboration (CKD-EPI) is considered the most precise.
The new Japanese coefficient for eGFR based on insulin clearance data has been shown to be more accurate for the Japanese population than the previously reported equations.
Serum cystatin C has been evaluated as an alternative or additional renal biomarker for estimating kidney function. However, the serum cystatin C level can be influenced by:
- age
- sex
- race
- other non-renal factors
Might be useful in early identification of tubular damage, especially in patients receiving HAART, is the periodic monitoring of urinary low molecular weight proteins, such as:
- N-acetyl-β-d-glucosaminidase
- β2 microglobulin
- α1 microglobulin
In the pre-HAART era, dialysis was not offered to patients with HIV infection because of poor survival and with high infection rates.
Currently, both peritoneal dialysis and hemodialysis are effective modes of renal replacement therapy in these patients.
Prior to the introduction of HAART, the morbidity and mortality of HIV-infected patients was too high to justify using scarce resources to transplant HIV-infected patients.
The ability to suppress HIV replication with HAART, as well as improved prophylaxis and treatment of opportunistic infections, encouraged the transplant community to select patients with HIV to be included on the transplant list.
Conclusion
All HIV-infected persons should be screened at regular intervals for a history of:
- metabolic disease
- dyslipidemia
- diabetes
- hypertension
- alteration of body composition
Cardiovascular risk and renal function should also be assessed.
The IDSA guidelines recommend biannual screening for proteinuria, glycosuria, eGFR, and phosphateof patients on tenofovir with eGFR<90 mL/min, concomitant boosted protease inhibitors usage, or renal risk factors.
Referral should be considered in patients with:
- decreasing eGFR
- lingering proteinuria
- hematuria
- pyuria
Adaptation of dosages might be required in the case of more drugs.
Kidney biopsies are commonly recommended to ascertain an accurate diagnosis, except contraindications.
To reduce dyslipidemia, lifestyle interventions should focus on counseling to:
- stop smoking
- follow a healthy diet
- restrict salt intake
- exercise
- maintain normal body weight
If not effective, a change in HAART should be considered, followed by the use of statins.
A pre-emptive switch from thymidine analogues is recommended to reduce the risk of development or progression of lipoatrophy.
Intra-abdominal fat accumulation is best managed by exercise and diet, in cases where lipoatrophy has developed, reversal is slow and gradual.
Prevention and management of diabetes and hypertension follow guidelines used in the general population.
When using medical interventions to prevent and/or treat metabolic diseases, impairment of the efficacy of HAART should be prevented by considering the possibility of pharmacokinetic interactions and compromised adherence.
Treatment of HCV co-infection should be considered when patients have HCV-related nephropathy proved by renal biopsy.
Still, the safety and efficacy of interferon treatment for HCV infection in HIV-infected individuals has not been documented.
For a broader and deeper understanding of nephropathies in HIV-infected patients, specialists in HIV and experts in metabolic diseases should consult each other.

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles