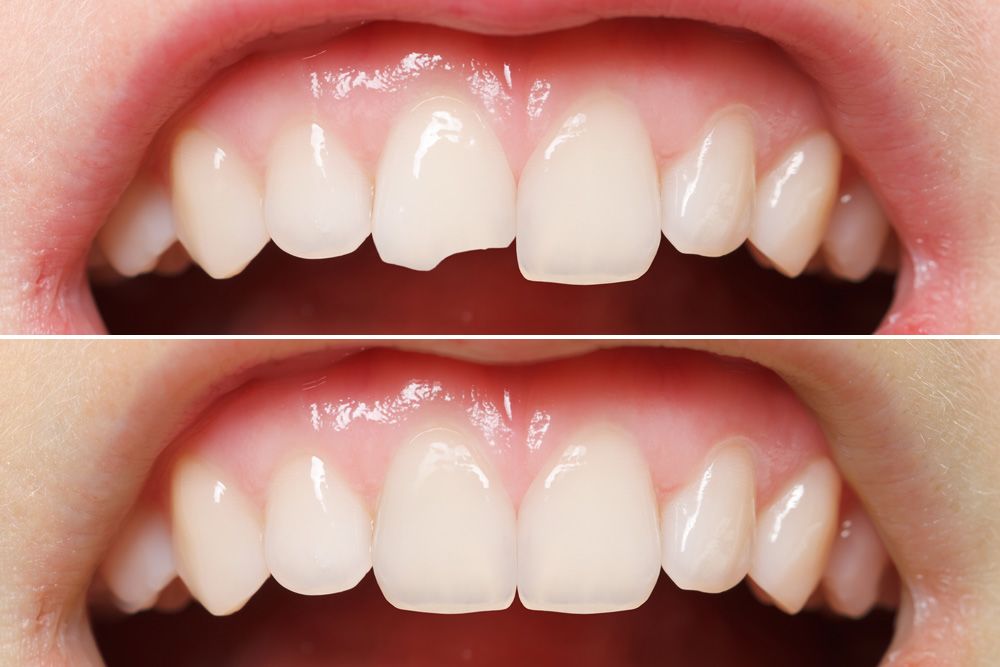
Dental Bonding - How It Has Aided In Teeth Restoration
Author:Suleman ShahReviewer:Han JuAug 06, 202222 Shares448 Views

When our teeth become damaged due to trauma or dental caries, we are required to employ bonding materials or filling materials. This is something that we are all well aware of.
Many advancements and improvements have been made to dental materials over the course of time.
Our goal is to explain how the use of these materials has resulted in enhanced dental healthand restoration.
The term "adhesion" refers to the process in which two surfaces attach to one another. Dental bondingis determined by the quality of the component parts, which include the tooth, the prosthesis, and assembly materials such as cement and glue.
There are two primary types of adhesion that can be distinguished from one another: mechanical and chemical.
The principle behind mechanical adhesion is called mechanical interlocking, and it serves to give both retention and a long-lasting form of bonding.
To achieve chemical adhesion, the surface etch must first be modified, and then two surfaces that are quite different must be joined together using an active monomer.
What Is Dental Bonding?
Bonding your teeth is a cosmetic dentistry treatment that can be used to improve the appearance of your smile. It is also known as composite bonding or tooth bonding. During the process, your dentist will apply a material that is tooth-colored resin to the affected teeth in order to change the shape, size, or color of the teeth.
What Benefits Can Dental Bonding Offer?
When compared to other aesthetic treatments, cosmetic dental bonding has numerous advantages to offer patients. For instance, the procedure is as follows:
Minimally invasive: Dental bonding, on the other hand, often does not call for the removal of enamel from the tooth, in contrast to procedures such as porcelain veneers and dental crowns.
Cost-effective: Dental bonding is one of the cosmetic dental procedures that is one of the least expensive options available.
Versatile: Dental bonding has the ability to cover a wide variety of cosmetic flaws, such as chips, cracks, gaps, and discoloration.
Fast and convenient: Other aesthetic operations, such as veneers and crowns, need to be performed over the course of several appointments. The dental bonding process can be completed in just one visit to the office.

Cosmetic Bonding
What Are The Drawbacks Of Dental Bonding?
Although dental bonding material is moderately stain-resistant, porcelain restorations are more stain-resistant than dental bonding material. Another drawback of bonding is that it doesn't last as long as other restorative treatments like veneers or crowns. Additionally, with time, bonding materials may chip.
Dental bonding is a great option in general for minor cosmetic adjustments. Other procedures, including porcelain veneers, might be a better fit for you if you're hoping to see a more dramatic improvement.
Dental Assembly Materials
Cement
The processes used for curing cement or glue largely characterize these substances. Materials for the acid-base reaction cure in cement. Alkaline powders and acidic liquids are the two most typical forms.
The liquid is made up of a copolymer of acrylic acid and other unsaturated carboxylic acids, or an aqueous solution of polyacrylic acid. The powder is fluoroaluminosilicate glass. Their characteristics are determined by the types of liquid and powder.
Due to its poor mechanical qualities, high cytotoxicity, and high solubility, silicate cement is not used. The least cohesive substance is zinc polycarboxylate. They are utilized less because of their weak mechanical qualities. Glass ionomer cement has a somewhat lower compressive strength than zinc orthophosphate.
Both glass ionomer cement (GIC) and zinc polycarboxylate cement (ZPC) can release fluoride and give direct adhesion to the tooth when used in dental applications, but ZPC is more radiopaque and biocompatible than GIC.
An acid-base interaction also determines how polycarboxylate cement sets. Zinc oxide (ZnO), magnesium oxide (MgO), bismuth (Bi2O3), and aluminum oxide make up the majority of the powder's ingredients (Al2O3). Additionally, it could contain stannous fluoride (SnF2), whose disappearance enhances the orthodontic bracket's shear bond strength (SBS).
Because of its superior mechanical strength and biocompatibility, zinc oxide (ZnO) particles have been employed for temporary and permanent luting cement. ZnO nanoparticles have a high surface-to-volume ratio, unusual optical characteristics, and antimicrobial capabilities.
Adhesives
The adhesive is primarily an organic polymer that includes a methacrylate matrix, where the matrix may or may not contain mineral filler. It is made up of liquid and powder.
Two monomers of methacrylic acid are used to create the resin matrix. The most popular ones are methyl methacrylate (MMA), bis-GMA, and UDMA. To lessen the high viscosity produced by bis-GMA, ethylene glycol (TEGDMA) derivatives are typically added.
The filler contains sub-micron sized colloidal glass silicon particles, such as SiO2, B2O3, Na2O3, and Al2O3 (particle size: 10–40 nm). They make about 30% to 65% of the glue volume. The volume ratio of the matrix to the filler has a significant impact on the adhesive characteristics.
As a result, increasing this ratio will result in less polymerization removal and more viscosity. A robust chemical link between the matrix and the filler is necessary for the formation of a cohesive structure. Otherwise, every interface will serve as the prime site for the start of cracks.
The polymerization initiator starts the redox process, which is followed by the curing phenomena. Always follow the redox reaction before the beginning. Free radicals produced by the process attack the methacrylic group's double bond, causing polymerization. An amine peroxide pair typically initiates the adhesive's chelating process.
Dental Bonding Video | Tooth Bonding
Adhesion To Teeth
Contrary to popular belief, most adhesives do not adhere to surfaces. Due to active monomers like 4-methacryl ethyl triacrylate (4-META) and 10-methacryl decyl phenyl phosphate(10-MDP), only a select few adhesives are able to adhere.
A coupling agent is therefore employed to adhere to the dental surface. A reactive polar group in the coupling agent can be coupled to oral tissue on the one hand and an adhesive substance on the other (HEMA).
Adhesion To Enamel
The very hydrophobic traditional resins are incapable of adhering to the dental surface.
The answer to bonding these surfaces is to identify an adhesive, a coupling agent to bind to the tooth tissues, and an advantageous surface topology for anchorage.
The enamel surface can be cleaned by phosphoric acid etching, which can also increase the substrate's surface energy and the adhesive's wettability. Acid etching, on the other hand, creates a roughness (up to 50 microns) where resin can pierce. In actuality, the composite structure of this highly mineralized tissue results in the selective etching of particular hydroxyapatite phases.
Dentin Adhesion
Due to the complexity of dentin's structure, bonding with dentin presents a number of challenges.
- Dentin mud covers the dentin. It should be remembered that milling forms a deposit smearing layer when dentin is removed.
- The depth of the tubule affects the complexity of its structure. In actuality, only 1% of the dentinal tube is present on the tooth surface near the dentinoenamel junction, while 22% of the dentinal tube is present close to the pulp.
- Mineral phase (70%), organic phase (20%).
- Dentin continues to form throughout life.
- Dentins are hydrophilic and saturated with oxygen.
Therefore, these obstacles make it difficult to develop reliable dental adhesive systems.
Bonding System Application
Contrary to popular belief, most adhesives lack the ability to stick to surfaces. Due to their active monomers, only a select few adhesives, like 4-methacryl ethyl triacrylate (4-META) and 10-methacryl decyl phenyl phosphate (10-MDP), have this capability.
A coupling agent is therefore employed to adhere to the dental surface. A reactive polar group in the coupling agent can be coupled to oral tissue on one end and adhesive material on the other end (HEMA).
Because the great majority of adhesives lack adhesive properties, bonding is accomplished by creating a mixing layer between the porcelain or teeth and the glue (HEMA).
However, there are adhesion-capable active monomers such (4-META, 10-MDP, and 11-methacrylic oxygen-1, 1-eleven alkane two carboxylic acids (MAC 10).
Measurement Of Bond Strength
Shear and tensile tests are common for testing bond strength. Regardless of the test, interface shape, force loading, and test speed affect bond strength.
This diversity, which makes comparing research difficult, may be attributed to the interface's geometry, force loading, and test solicitation speed. Dental adhesion tests cannot separate the impact of adhesive composition, substrate parameters, joint geometry, and loading type on determined bond strength.
Adhesion energy tests are used to evaluate an adhesive joint's material qualities. These tests involve initiating and propagating a crack through a bonded contact.
Double cantilever beam (DCB) and notchless triangular prism (NTP) tests can evaluate a bonded joint's adherence energy. Theoretically, these tests yield a genuine, characteristic value of bonding joints irrespective of specimen geometry.
A prior study outlined the DCB's mechanics and principles. The DCB test evaluates Gc, which shows the metal/resin interface's durability.
DCB reveals how microstructure increases dental composite toughness. The DCB test was recently used to analyze the mode I fracture energy of hydrated and thermally dehydrated bovine femur cortical bone, which is identical to human dentine. Clinical findings are corroborated by DCB test results.
People Also Ask
Is Dental Bonding Painful?
Because the dentist only works on the tooth's surface most of the time, the process of placing dental bonds does not in any way cause discomfort to the patient. If your bond is used to repair a cavity, then you may feel some pain throughout the procedure.
How Much Does Dental Bonding Cost?
You should budget between $300 and $600 on average for each tooth. Every five to ten years, the bonding will need to be replaced. Before making an appointment, check with your dental insurance company. Certain insurance companies do not cover dental bonding because they view it as a cosmetic procedure.
Is Dental Bonding Worth It?
It is possible for people to find successful solutions to their cosmetic or dental health problems. Bonding is an option to consider if you are seeking a solution to problems such as chipped teeth, discolored teeth, or mildly damaged teeth.
Does Dental Bonding Look Natural?
The answer to the question of whether or not tooth bonding looks natural is that it does. The dental bonding substance has been properly created, and its color has been matched to the natural teeth, so that it looks completely natural. Unless you choose to draw attention to the fact that you have a bonded tooth, the majority of people won't notice that you have one.
Conclusion
Despite the fact that dental bonding systems are getting easier to use and more effective, dentists still need to stick to the precise sequence of steps, observe the proper application times for each step, and follow the directions provided by the manufacturer. Dentists should be aware that not all structures are the same and that not all materials, whether composite, porcelain, zirconia, or metal, can be treated in the same manner, despite the fact that fewer bottles are required.
It is envisaged that in the field of dental bonding, in the future, a greater number of dentists will be able to maintain their patients' natural teeth and bring the research on bonding materials up to speed. It is possible that the cutting-edge technologyof dental bonding materials may provide a significant boost to the field of study concerning dental bonding.

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles