Gliomas - The Effects Of Advanced Imaging Techniques
Gliomas are the most frequent primary brain tumors, which have a range of prognoses based on their grade and genetic makeup. Patients often undergo imaging testing, frequently an MRI scan, which identifies the neoplastic lesion in patients who first come with seizures and/or a localized neurological disability.
Author:Suleman ShahReviewer:Han JuJul 29, 2022133 Shares2.1K Views
Gliomasare the most frequent primary brain tumors, which have a range of prognoses based on their grade and genetic makeup.
Patients often undergo imaging testing, frequently an MRI scan, which identifies the neoplastic lesion in patients who first come with seizures and/or a localized neurological disability.
Maximal safe resection is the first step in the standard of treatment for these patients, which is then followed by chemotherapy and radiation.
There is strong evidence that the maximal extent of resection (EOR) of the contrast-enhancing (CE) tumor regions on post-contrast T1 imaging, as well as in some cases, the resection of non-contrast enhancing (NCE) disease that extends beyond the CE tumor (i.e., a supratotal resection), improves survival in the case of high grade glioma (HGG).
Advanced imaging modalities, such as perfusion imaging, diffusion imaging, spectroscopy, and positron emission topography (PET) imaging, have emerged as increasingly useful surgical adjuncts for achieving a maximally safe resection of these infiltrative tumors, particularly when the goal is a supratotal resection. This is especially true when combined with intraoperative brain mapping.
However, despite intensive therapies, these cancers nearly invariably come back, and early tumor recurrence diagnosis is still essential for the best patient care and assessment of available treatment choices, including the viability of further surgical resection.
What Are Gliomas And Types?
A particular kind of tumor known as a glioma develops in the brain and spinal cord. The glumpy support cells (glial cells), which surround and support nerve cells, are where gliomas develop.
Glial cells may become tumors in three different ways. The genetic characteristics of the tumor, which may assist in anticipating how the tumor will behave over time and the therapies that are most likely to be effective, are used to classify gliomas.
Glioma types include:
- Oligodendrocytes, a kind of glial cell, are the origin of oligodendroglioma tumors. Although oligodendrogliomas often develop more slowly, they might eventually become more aggressive. They seldom spread beyond the brain or spine, much like ependymomas. Adults experience them more often than kids do. Oligodendrogliomas account for about 1% to 2% of all brain tumors.
- Ependymocytes, a kind of glial cell, are the origin of ependymoma tumors. Ependymomas often develop in the spinal cord or brain ventricles. The fluid that surrounds and protects the brain and spinal cord, known as cerebrospinal fluid, may allow them to spread, but they cannot leave the brain or spinal cord. Approximately 2% of all brain tumors are epidermoid. Children experience them more often than adults.
- Glioblastomas and diffuse intrinsic pontine gliomas (DIPGs), among other astrocytomas: These cancers develop from astrocyte-like cells. Astrocytomas that develop quickly or with extreme aggressionare known as glioblastomas. The majority of adult malignant brain tumors are these. In youngsters, gliomas and astrocytomas are frequent. DIPG is an extremely uncommon but severe kind of pediatric brain cancer. It typically affects youngsters and develops in the brain stem.
Depending on where it is and how quickly it is growing, a glioma may impair your brain's function and pose a danger to your life.
Gliomas are one of the most prevalent kinds of primary brain tumors.
Your prognosis and course of therapyare influenced by the kind of glioma you have. Surgery, radiation therapy, chemotherapy, targeted therapy, and experimental clinical trials are the main glioma treatment options.

Glioma: Symptoms, Diagnosis & Treatments
What Are Some Symptoms Of Gliomas?
Possible symptoms of gliomas are as follows:
- Seizures.
- Personality or behavioral changes.
- Nausea and vomiting.
- Hemiparesis (symptoms include numbness or weakness on one side of the body).
- Headaches.
- Dizziness.
- Walking or maintaining your balance may be difficult for you.
- Cognitive problems (having difficulty concentrating, learning, or remembering).
- Vision impairments or abnormalities might be occurring.
- Aphasia (issues in articulating one's thoughts and communicating).
Preoperative Imaging And Surgical Planning
Most institutions use a defined methodology for lesion characterization and pre-operative planning if an intra-axial brain tumor is diagnosed. These methods include 3-dimensional (3D) T1 pre and post-contrast-enhanced, T2 contrast-enhanced, fluid-attenuated inversion recovery (FLAIR), and diffusion-weighted imaging (DWI) at a magnetic field strength of 1.5 tesla (T).
In the next part, we detail these typical sequences and how they might be utilized to clarify the diagnosis, evaluate tumor invasion, and optimize resection safety.
T1 Pre And Post-Gadolinium And T2/T2 Fluid-Attenuated Inversion Recovery Sequences
T1-weighted pre and post-contrast pictures as well as T2 weighted images, especially the fluid-attenuated inversion recovery (T2 FLAIR) sequence, are typically most crucial for tumor visibility and are used intra-operatively with neuronavigation to help in tumor excision.
High grade gliomas induce blood-brain barrier (BBB) rupture and vascular leakage of gadolinium contrast agent. Extravasation of contrast agents causes T1 shortening and hyperintensity on T1-weighted imaging. Malignant cells infiltrate T1 contrast-enhanced regions in glioblastoma (GBM).
A recent multicenter cohort study investigating maximal resection of CE and NCE tumors showed that in addition to the survival benefit conferred by maximal resection of the CE tumor, additional resection of the NCE tumor improves overall survival (OS) irrespective of isocitrate dehydrogenase (IDH) and O6-methylguanine-DNA methyltransferase (MGMT) methylation status in younger patients. This highlights the need for imaging methods to delineate tumor infiltration beyond high contrast enhancement.
Diffusion-Weighted Imaging
Diffusion-weighted imaging (DWI) measures random (Brownian) water molecule movement and calculates diffusion metrics such as apparent diffusion coefficient (ADC). In glioma imaging, limited diffusion (low ADC signal) implies hypercellularity owing to significant tumor growth and water diffusion restriction.
Hypercellular regions restricting water diffusion look bright on DWI. Studies show an inverse association between ADC and cellular density and tumor growth.
Intratumoral heterogeneityand necrosis may restrict ADC readings in tumor locations. Hypercellularity may identify early phases of malignant transformation that don't exhibit contrast enhancement on T1.
Susceptibility-Weighted Imaging
Susceptibility-weighted imaging (SWI) is an MR sequence that exhibits changes in the local magnetic field susceptibility between surrounding tissues, allowing for the observation of substances such as deoxyhemoglobin in venous blood, iron deposition in the brain, or calcium. SWI pictures are formed by processing high-resolution (3.0 T), long echo time (TE), full-flow correction, and 3D gradient-echo data.
The MR data is analyzed to identify ferromagnetic (iron), paramagnetic (deoxyhemoglobin, clots), and diamagnetic compounds in tissues (calcium). Imaging shows diamagnetic calcium as brilliant and paramagnetic blood as black.
SWI is the ideal technique for imaging microhemorrhages due to its ability to discriminate necrosis from bleeding following radiation therapy. SWI's capacity to detect calcium on imaging may help forecast tumor histology and/or grade before pathological confirmation, especially if a CT scan is not available.
In oligodendrogliomas, gangliogliomas, pilocytic astrocytomas, and ependymomas, intratumoral calcification appears as low signal intensity. SWI may assist in identifying brain abscess and necrotic GBM when T1 post-contrast imaging is problematic.
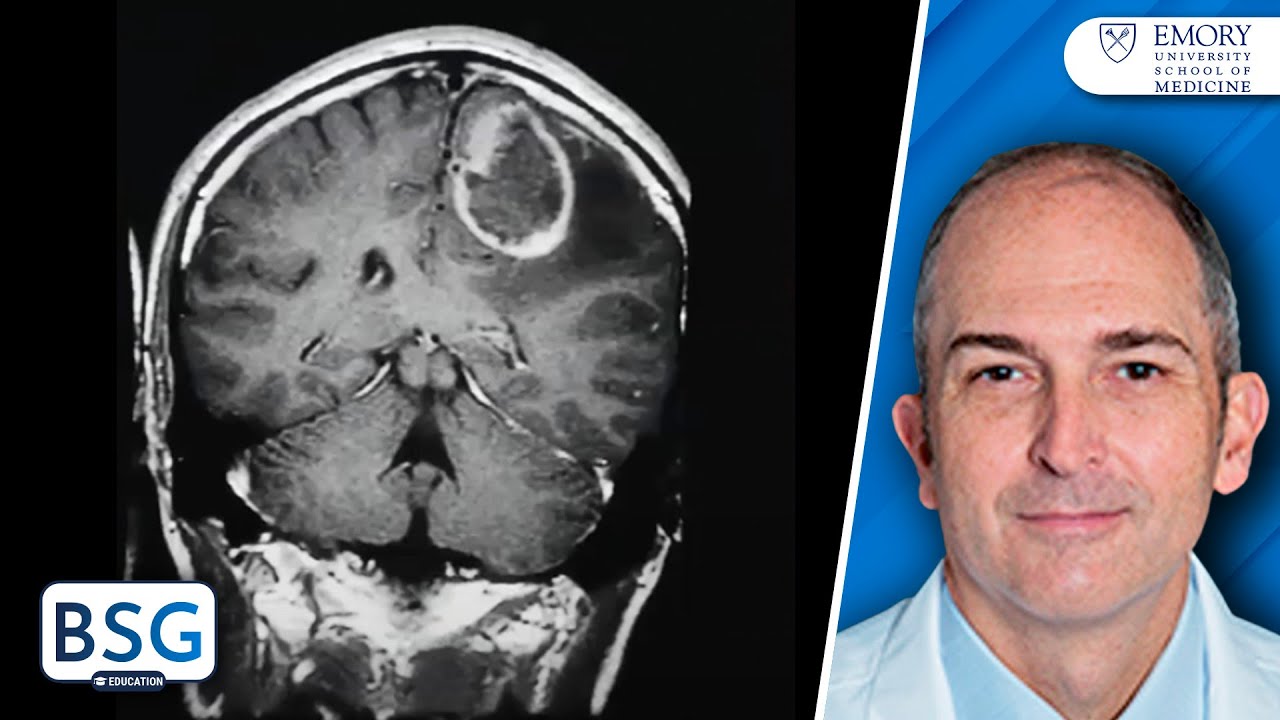
Glioma Classification, Diagnostics, and Treatment
Perfusion MR Imaging
Clinically, dynamic susceptibility contrast (DSC), dynamic contrast enhanced(DCE), and arterial spin labeling (ASL) are the most prevalent MR perfusion methods. DSC measures the transitory reduction in brain signal intensity on T2*-weighted sequences following gadolinium contrast agent delivery to construct a signal intensity time curve used to calculate rCBV for each voxel.
The rCBV signal correlates directly with microvessel volume and is a measure of angiogenesis, distinguishing HGG from LGGs and non-neoplastic etiologies such as post-treatment impact with larger rCBV signal in higher grade gliomas.
This modality is predictive of malignant transformation of LGGs and overall survival, and it can help distinguish tumor recurrence from post-treatment effects such as radiation necrosis and pseudoprogression, defined as a transient increase in post-contrast enhancement within the treated tumor lesion that presents during the first 6 months after treatment, followed by spontaneous radiographic improvement or resolution.
MR Spectroscopy
MR spectroscopy (MRS) helps define brain areas' biochemical makeup. This comprises steady-state metabolite concentrations, metabolic response rates, and cellular transport.
MRS reveals variations in normal and malignant biochemical makeup. MRS provides insight into cell membrane turnover (Cho), neuronal vitality (NAA), normal cellular metabolism (Cr), tissue hypoxia (Lac), and astrocytic integrity (MI) by measuring the presence of specific metabolites such as choline, N-acetylaspartate, creatinine, lactate, and myo-inositol (MI).
Cho/NAA and Cho/Cr metabolite peaks correlate positively with tumor grade and may discriminate vasogenic from infiltrative edema. Emerging MRS techniques may detect 2-hydroxyglutarate (2-HG), an oncometabolite generated by IDH mutant tumor cells, as a non-invasive evaluation of LGG-uncertain lesions.
Histologically verified studies suggest 2-HG detection using MRS can identify IDH mutant from IDH wildtype glioma with varied sensitivity and specificity. This modality's limitations include the necessity for a significant lesion inside a voxel, lack of technical standards, and volume averaging.
Positron Emission Tomography As A Tool For Preoperative Planning
Positron emission tomography (PET) measures cell proliferation, glucose metabolism, amino acid absorption, and membrane production using radio-labeled tracers. This neuro-oncology clinical tool delivers molecular, functional, and metabolic tumor biology information.
The most common molecular tracers utilized for PET imaging of gliomas include those that image glucose metabolism—2-18F-fluoro-2-deoxy-D-glucose (18F-FDG)—and those that image amino acid transport—O-(2-18F-fluoroethyl)-L-tyrosine (18F-FET), (S-11C-methyl)-L-methionine (11C-MET), and 3,4-dihydroxy-6-18F-fluoro-L-phenylalanine (18F-FDOPA).
PET imaging with 18F-FDG compares tumor uptake to normal brain parenchyma. 18F-FDG PET uptake correlates with tumor grade and survival.
18F-FDG PET can discriminate between GBM and CNS lymphomas during initial tumor diagnosis due to their different 18F-FDG absorption rates. 18F-FDG PET for glioma diagnosis has limitations.
Normal cerebral cortex glucose metabolism and 18F-FDG absorption restrict 18F-FDG PET accuracy. This modality can't discriminate between gliomas, metastatic lesions, and non-neoplastic lesions such as brain abscesses, neurosarcoidosis, and some demyelinating CNS illnesses. In presurgical planning, amino acid PET tracers are recommended over 18F-FDG.
Imaging For Monitoring Treatment Response And Tumor Recurrence
Despite intensive therapy, virtually all diffuse gliomas relapse. Postoperative imaging is crucial to detect tumor recurrence as early as possible to reduce disease development. Despite gross complete tumor excision, numerous studies demonstrate substantial LGG recurrence rates within 5 years.
Early detection of LGG tumor recurrence reduces the risk of malignancy. GBM recurrence is inevitable and hard to control. Bevacizumab, nitrosourea alkylating drugs, and/or repeat tumor resection are used to treat recurrent GBM.
Postoperative monitoring for tumor recurrence and treatment-related alterations is difficult since glioma recurrence and gliosis or treatment effects seem identical on T1 and T2 sequences. A hyperintense signal on T1 post-contrast imaging inside the resection cavity in the postoperative phase is alarming for tumor recurrence but may potentially indicate ischemic brain tissue or devitalized tumor.
Inflammatory processes after chemoradiation may contribute to temporary edema that can resemble tumor recurrence for months in HGG and LGG patients. This is commonly misinterpreted as tumor advancement but is really pseudoprogression.
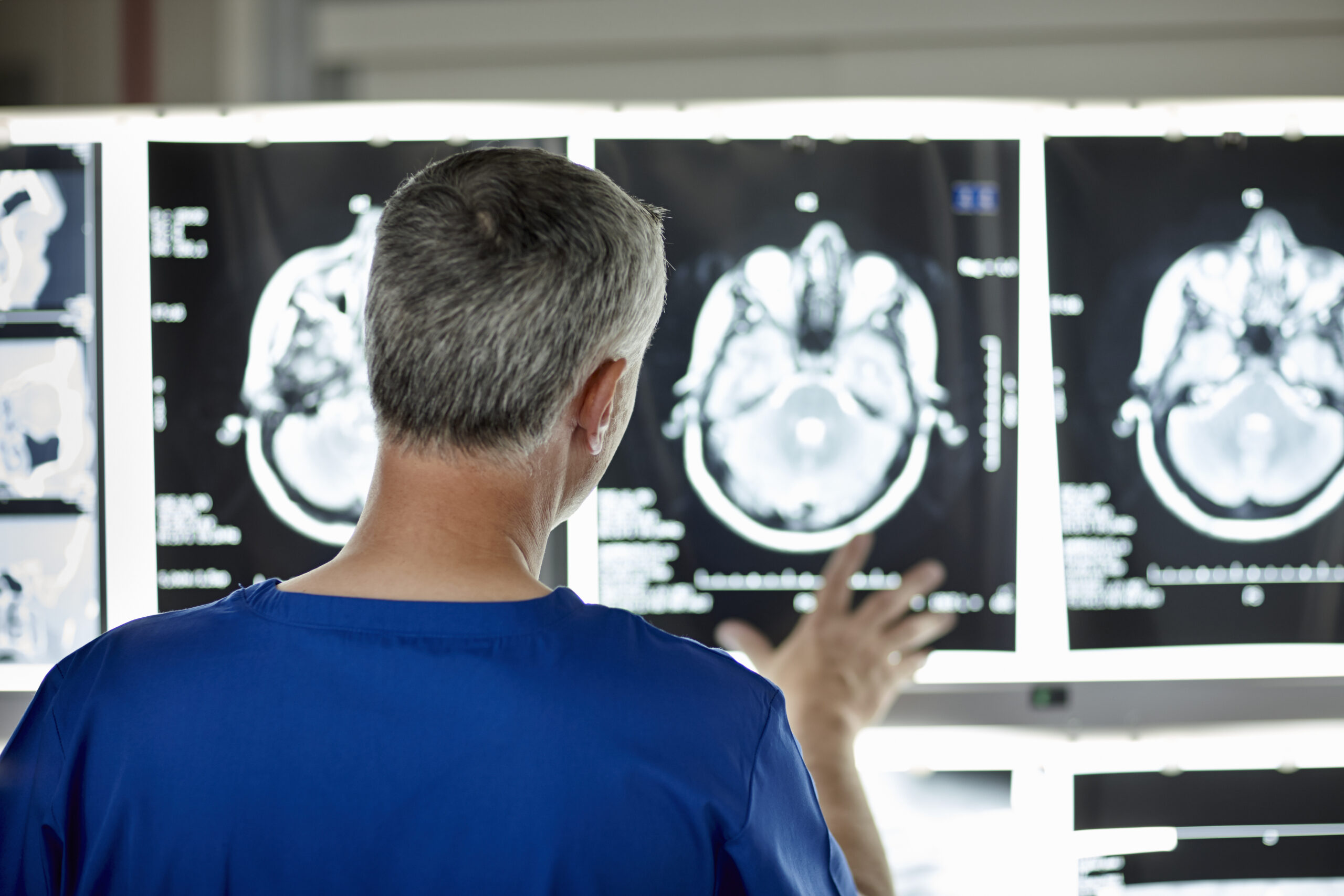
Imaging To Assess For Pseudoresponse Following Immunotherapy
Pseudoresponse is a reduction in contrast extravasation owing to BBB leakiness, resulting in reduced CE on T1 post-contrast imaging. This occurs after antiangiogenic therapy with bevacizumab.
Bevacizumab-treated GBM recurrence patients. The anti-VEGF-A characteristics of bevacizumab led to a quick radiologic response of contrast enhancement reduction and reduced edema days after therapy. However, current research suggests bevacizumab does not improve OS.
This is likely owing to tumor adaptation to antiangiogenic treatment, resulting in a hypoxic NCE invasive tumor phenotype that may survive despite diminished vascular growth. As a result, researchers are developing a generalizable method for detecting pseudoresponse in GBM.
Emergence Of Radiomics
Radiomics is the art of collecting and interpreting quantitative information from diagnostic pictures to create prognostically relevant information such as treatment response vs. tumor progression and survival projections.
Radiomics may characterize heterogeneity over the whole tumor volume, while tumor biopsies only capture local heterogeneity. Existing radiomic approaches have been used to analyze intratumoral features in GBM.
One study gathered image-guided regional GBM samples, identified regional GBM driver genes, and co-registered biopsy locations with MRI and texture maps to link genetic status with imaging parameters. Machine learning methods were used to detect MRI characteristics at the voxel level that corresponded with GBM driver gene status. This approach may build an imaging algorithm that captures tumoral molecular markers based on stereotyped imaging patterns.
Radiomics And Recurrence
Wang et al. have developed a radiomics model to distinguish glioma tumor recurrence from radiation necrosis. This model was created in 112 patients with pathologically diagnosed gliomas and validated in 48 more cases.
18F-FET PET, 11C-MET PET, and patient data were used to predict tumor recurrence. The integrated model, which included 15 significant indicators of recurrence (p < 0.001) such as mean tumor-background ratio (TBR) of 18F-FET, maximum TBR of 11C-MET, and patient age, was highly predictive of recurrent glioma and accurate across both test and validation populations.
More work is needed to confirm radiomics-based models are generalizable across institutions with different scaling parameters, but this is an exciting emerging field that is advancing glioma understanding and developing non-invasive prognostic information. These are critical first steps in enhancing tailored patient care for glioma treatment. However, more work remains.
People Also Ask
Why Do Gliomas Occur?
Glial stem or progenitor cells accumulate genetic alterations over time, which causes their unchecked proliferation and results in gliomas. Mutated genes often play a role in processes including DNA repair, tumor suppression, and cell growth control.
Can Gliomas Be Cured?
Gliomas with a high grade cannot be healed. Concerns about the patients' quality of life are an extremely important factor to take into consideration while treating high-grade glioma patients.
Where Are Gliomas Usually Found?
The cerebrum (the vast, outer portion of the brain) and the cerebellum are where they are most often found (located at the base of the brain). Children or adults may develop astrocytomas. Glioblastoma multiforme, also known as high-grade astrocytomas, is the most dangerous type of all brain tumors.
Are Gliomas Hereditary?
Because glioblastomas are so aggressive, they often develop and spread quickly. Glioblastomas develop randomly in individuals without a personal or familial historyof brain malignancies. Therefore, scientific data points to the large majority of situations in which this cancer is not inherited.
Conclusion
The treatment of glioma patients has been transformed by the use of MR and PET scans. T1, T2/FLAIR, DWI, and susceptibility images are often obtained after the diagnosis of an intra-axial brain tumor in order to aid in preoperative planning. The utilization of cutting-edge imaging technologies, including MR perfusion, diffusion, spectroscopy, and PET scans, is increasingly being used to further improve this treatment.
Additionally, to increase the safety of glioma resection, functional imaging evaluations, intra-operative use of anatomical images, and neuronavigation are all utilized. Serial imaging is crucial for tracking cancers throughout therapy, but it may be difficult to tell when a tumor is responding to the treatment and when it is progressing.
Standardizing the use of procedures like MR perfusion, diffusion, and spectroscopy as well as PET scans for postoperative tumor monitoring will be crucial in the future as they may significantly enhance interpretation of the underlying biologic process.
Last but not least, radiomics is developing into an intriguing big data tool for quantifying the information presented in the pictures, possibly boosting precision and accuracy, although its applicability and significance in the treatment of gliomas are yet unclear.

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles