Human Aquaporins - Their Effect On Infectious And Non-infectious Diseases
Human aquaporins are integral membrane proteins that make channels and are found in all living things, like bacteria and chlorella viruses. Aquaporins (AQPs) are proteins that allow water and other small solutes to move through cell membranes. They are also involved in cell trafficking and a number of other physiological functions.
Author:Suleman ShahReviewer:Han JuJul 23, 20229 Shares380 Views
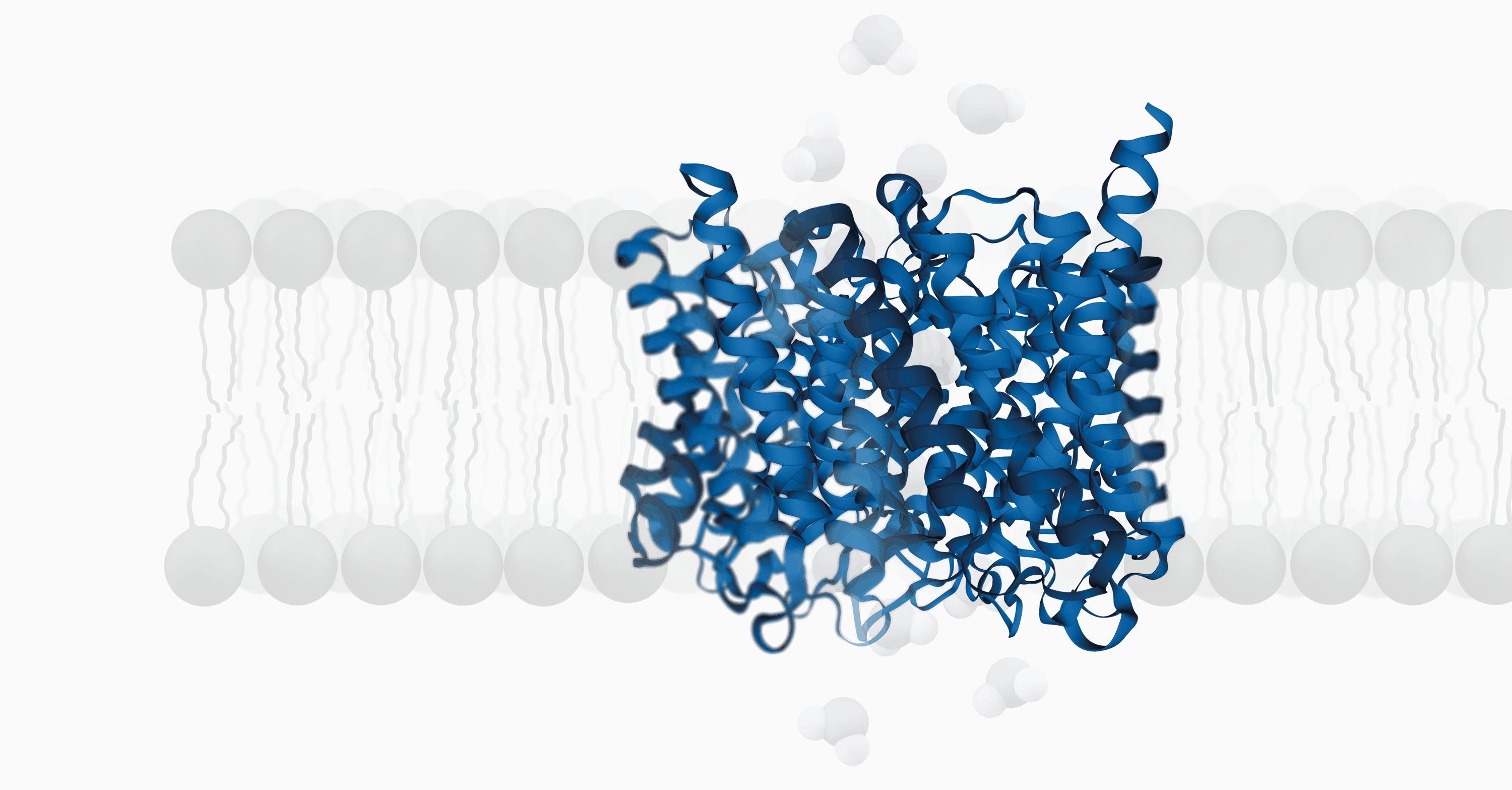
Human aquaporinsare integral membrane proteins that make channels and are found in all living things, like bacteria and chlorella viruses.
Aquaporins (AQPs) are proteins that allow water and other small solutes to move through cell membranes. They are also involved in cell trafficking and a number of other physiological functions.
Human AQPs with functionally divergent functions are implicated in a broad range of non-infectious disorders, including cancer, renal failure, neurological problems, epilepsy, skin disease, metabolic syndrome, and even cardiac diseases.
Growing evidence suggests that they may be involved in cell volume regulating processes linked with a variety of non-infectious illnesses. As a result, AQPs have emerged as a possible therapeutic target in clinical medicine.
Much research and reviews have been published on the role of AQPs and their regulation in non-infectious illnesses.
However, few investigations into AQPs in infectious illnesses have been conducted, despite advances in plant-pathogen interactions involving AQPs.
Infectious diseases, which are illnesses caused by bacteria, viruses, fungi, or parasites, are among the leading causes of morbidity and death in people all over the globe.
Structural Properties Of Human Aquaporins
Despite differing amino acid sequences, AQPs have a highly conserved tetrameric structure; each subunit functions as a channel. The tetramer's core forms a fifth pore.
Each monomer contains six transmembrane (TM) α-helices (H1-H6), five connecting loops (loops LA-LE), and cytoplasmic N and C-termini, which combine to form a pore that defines transport activity.
The canal has two bottlenecks. The first constriction is created by two proton-excluding Asn-Pro-Ala (NPA) motifs on loops B and E. Both NPA motifs extend into the membrane and create the seventh pseudo TM helix.
Four residues from helix H2 and H5, and loop E (LE1 and LE2) create the aromatic/arginine (ar/R) selectivity filter. This ar/R selectivity filter may influence substrate conductivity.
All AQPs have the same core structure, but their loops and N and C ends are different, which shows that they have different functional and/or regulatory roles.

The Aquaporin Inside® Technology Explained
Human Aquaporins Have Tissue-Specific Distribution And Physiological Relevance
A summary of tissue-specific AQP distribution and physiological importance AQP1–11 isoforms are expressed in the brain and nervous system. AQP1 and AQP4 have increased brain expression. AQP1 helps produce cerebrospinal fluid (CSF) in choroid plexus epithelial cells.
AQP1 is found in primary sensory neurons and may affect pain perception. AQP4 is present in astrocyte cell membranes, ependymal cells, and hypothalamic osmosensory regions. Eight of thirteen human AQPs are in the eye (AQP0, 1, 3, 4, 5, 7, 9, and 11).
AQPs maintain corneal transparency, corneal wound healing, tear film osmolarity, and retinal homeostasis. In the human ear, nine AQPs (AQP1, 2, 3, 4, 5, 6, 8, 10, and 11) regulate neuronal signal transduction, cell motility, lipid metabolism, and immune activities.
Six AQPs are present in the skin's epidermis (AQP1, 3, 7, and 10), dermis (AQP1, 3, and 5), and hypodermis (AQP7). AQPs affect skin moisture, cell proliferation and differentiation, migration, immunity, and wound healing.
AQP3 is involved in the growth and movement of epidermal cells. AQP1 controls the flow of water between the blood and dermis to keep the skin hydrated. AQP5, which is found in the epical and basolateral membranes of sweat glands, is involved in the secretion of water. AQP7 releases glycerol from adipocytes to make energy, and AQP9 moves glycerol, urea, and H2O2
Human Aquaporin Functional Diversity And Regulation
Aquaporins carry water and tiny solutes such as glycerol, nitrate, urea, ammonia, H2O2, CO2, O2, arsenic, antimony, silicon, sodium ion, and aluminum malate; some are physiologically significant and othersare hazardous heavy metals.
AQPs have critical physiological roles in humans, but mutations, malfunctions, and dysregulation cause many serious disorders. Transcellular water transport is set up by the number and activity of AQPs in cell membranes. Water molecules move along the osmotic gradient.
The thirteen human AQPs (AQP0-12) are classified into three types: orthodox AQPs (AQP0, 1, 2, 4, 5, 6, and 8), aquaglyceroporins (AQP3, 7, 9, and 10), and super aquaporins (AQP11 and 12).
Aquaporins are produced, inserted into the endoplasmic reticulum membrane, and localized to the target membrane through the Sec61 translocon. To maintain nutritional homeostasis in cells, their activity in the target membrane must be adequately controlled.
AQPs have many cellular and organ roles in humans. Some AQPs reside in intracellular vesicles but may go to PM. As a result of vasopressin and cAMP, AQP2 and AQP8 move from vesicles inside the cell to the PM.
AQP8 and 9 were found in the mitochondrial membrane. AQP trafficking targets PM for breakdown or recycling in the endosome.
Infectious Diseases - Aquaporin Roles In Infectious Diseases
Pathogenic bacteria, fungi, viruses, and parasites may cause systemic infections. Sepsis, the leading cause of death, is a clinical phenomenon that results from a local infection with an inflammatory response and has negative effects on the entire body, including organ malfunction.
Sepsis with two or more SIRS increases subsequent infection risk. Local and systemic infections may change how AQPs are expressed and how they work to keep cells and tissues in balance.
Video unavailable
This video is unavailable
Bacterial Infections And AQPs
Bacteremia or septicemia occurs when bacteria move into the circulation after infecting the skin, lungs, etc. This is dangerous because the bacteria's poisons may spread throughout the body through the bloodstream. Bacteria produce endotoxins and exotoxins that cause sepsis and death.
Bacterial endotoxins like LPS stimulate human AQP expression. Vassiliou et al. found that sepsis doubles AQP1 expression in ICU leukocytes (ICU).
Lipopolysaccharide (LPS) stimulates polymorphonuclear granulocytes (PMNs) for enhanced AQP1 expression and transiently increased cell volume during hypotonic therapy. LPS exposure raised AQP1 but reduced AQP3 and AQP5.
In human primary bronchial epithelial cells, LPS lowered AQP5, but not AQP3 or AQP4. Expression of AQP1 in the kidneys and heart may protect mice from AKI and cardiac hypertrophy caused by LPS and endotoxemia.
AQPs And Viral Infection
AQPs regulate cellular and tissue water homeostasis, which affects cell volume, migration, and organelle function. Multiple studies link AQPs to viral infections.
Human papilloma virus (HPV) infection alters AQP8 expression and functioning, causing male subfertility. AQP8 in sperm PM and intragranular membranes may enhance water and H2O2 transport, and AQP8-mediated H2O2 permeability may be implicated in ROS scavenging and detoxification. The interaction between HPV capsid protein L1 and AQP8 changes water and H2O2 permeability.
HPV causes cancer. HPV infection status and AQP1 and 3 overexpression are linked with poor cervical cancer outcomes, although their overexpression levels are not independent risk factors. AQPs may have changed the outlook for cervical cancer by making it easier for HPV to get into cells, grow tumors, and spread to lymph nodes.
Parasitic Infection And AQPs
Parasite taxa include protozoa, helminths, and arthropods. Leishmania, Plasmodium, and Toxoplasma are the most significant human protozoan parasites globally.
Parasites cause sickness or death in their hosts. The Plasmodium parasite infects erythrocytes via passing through liver hepatocytes. In the liver stage, the parasite matures into a merozoite and enters the bloodstream, causing malaria.
AQP helps transmit nutrients from the host to the parasite during the parasite's mandatory liver stage. A parasitophorous vacuole membrane (PVM) produced by the host to surround the parasite during invasion in hepatocytes and blood cells.
The PVM is needed for Plasmodium to grow. AQP3, which was found in the PVM, brought water and glycerol to Plasmodium and was needed for the parasite to grow in hepatoma cells.
AQP3 is recruited to the PVM in liver-stage P. berghei schizonts and blood-stage P. falciparum and P. vivax schizonts, and is assumed to enhance water or nutrition transfer between the parasites and the host cell. Infected hepatocytes showed considerably higher AQP3 expression than uninfected ones.
Involvement Of Aquaporins In Non-Infectious Diseases
Cancer, cerebral edema and ischemic stroke, renal failure, glaucoma, epilepsy, and obesity are linked to AQPs. Inherited NDI and congenital cataracts are caused by AQP gene defects.
AQPs In Cancer
Aquaporins are involved in cancer etiology, tumor edema, tumor cell migration and invasion, tumor proliferation, and angiogenesis. Twenty kinds of malignancies express AQPs.
Astrocytomas are infiltrating brain tumors that grow from astrocytes; the most aggressive grade IV is called glioblastoma. AQP1, 4, and 9 are highly expressed in human astrocytomas, whereas AQP1 is either up or down in cholangiocarcinoma. Colorectal, cervical, and breast malignancies up-regulate AQP1, 3, and 5, but down-regulate AQP8.
In liver cancer, AQP3 and 5 are higher than AQP8 and 9. Multiple studies link lung cancer to AQP1, 3, 4, and 5.
AQPs In Cerebral Edema And Ischemic Stroke
Cerebral edema plays a crucial part in the pathogenesis of many neurological illnesses, including ischemic damage, traumatic brain injury, brain tumors, and mortality. AQP4 in the end-feet of perivascular astrocytes controls the exchange of water between brain tissue and CSF in the ventricular and subarachnoid spaces.
In an ischemic stroke mouse model, AQP4 worsened post-ischemic cytotoxic edema, as evaluated by hemisphere enlargement, whereas AQP4-null animals had a better neurological prognosis. AQP4-knockout mice showed decreased mortality from hyponatremia in an acute water poisoning paradigm.
Absence of AQP4 may impair blood-brain barrier water permeability and brain water flow. AQP4 helps water to build up in the brain in cytotoxic edema and drain out of the brain in vasogenic and interstitial edema.
In a mouse model of acute cerebral ischemia, AQP4 expression was quickly up-regulated in perivascular end-feet, reaching the first peak after 1 hour (h) and the second peak in the penumbra after 48 h, corresponding with brain swelling. This shows that AQP4 is the main water route following brief cerebral ischemia.

Aquaporin
AQPs In Skin Diseases
Mutations, dysregulation, malfunction, or dysfunction of skin AQPs cause several skin disorders.
An inflammatory skin illness, erythema toxicum neonatorum, characterized by papules or pustules solely in neonates, is thought to be connected to elevated AQP1 in the epidermis, dermis, and blood vessels.
Recent research involving keratinocyte cell lines postulated that long-term exposure to blue light causes down-regulation of AQP1, 3, and 9.
Aquaporins As Potential Drug And Diagnostic Targets
All human organs exhibit aquaporins, which carry water, the necessary molecule for life, and other tiny molecules and ions. AQPs that don't work right, change, don't work right, or don't work at all are linked to life-threatening viral and non-infectious diseases.
Numerous infectious and non-infectious pathologies, including cancers, edema, obesity, brain injury, glaucoma, type 2 diabetes, NDI, AKI, CDK, DN, skin diseases, sepsis and sepsis-associated cholestasis, endotoxemia and endotoxemia-induced AKI, infective endocarditis, diarrhea, NMOSD, malaria, schistosomiasis,
AQPs are a pharmacological and diagnostic target. Numerous studies have suggested AQP-specific modulators and/or inhibitors, including heavy metal-based inhibitors, cysteine inhibitors, and small-molecule inhibitors, to modify or inhibit human AQPs and parasite AQPs.
Tissue and AQP-specific mechanisms regulate AQPs in infectious and noninfectious disorders. Infectious and non-infectious disorders should concentrate on the functional control of AQPs.
The regulation and functional functions of AQPs may provide novel insights into disease diagnosis and prognosis, as well as aid in the development of treatments.
Knowledge of AQP physiology is one method to overcome the limitations of present AQP modulators and inhibitors. Tissue-specific AQP distribution may need tissue or cell-specific AQP modulators.
Despite AQP's evident role in life-threatening disorders, especially infectious diseases, research on AQP-specific modulators is limited. AQP medication development might cure life-threatening disorders.
For AQP research to help solve problems in the real world, researchers must understand how the way AQP works affects how sick people get.
Changing where an AQP is located inside a cell would be a method that could be used for any future AQP-based drug development.
People Also Ask
What Are Examples Of Aquaporins?
Plasma-membrane intrinsic proteins (PIPs), tonoplast intrinsic proteins (TIPs), NOD26-like intrinsic proteins (NLMs or NIPs), and small basic intrinsic proteins (SIPs) make up the four main families of plant aquaporins.
Do Animals Have Aquaporins?
Aquaporins, or homologous water-channel proteins, are found in both plants and mammals. These channels are water-selective and block the passage of ions and metabolites.
How Many Types Of Aquaporins Are Found In Humans?
The 13 AQPs (AQP0-AQP12) found in humans can be divided into three subclasses based on how selectively they let water through their pores: orthodox aquaporins (AQP0, 1, 2, 4, 5, and 6), aquaglyceroporins (AQP3, 7, 9, and 10), and super or unorthodox aquaporins (AQP11 and 12).
Which Organ Would Require A Large Number Of Aquaporins?
Aquaporins are found in large amounts in many human organs, such as skeletal muscle and neurons that can't be excited by electricity, as well as the liver, kidneys, lungs, eyes, GI tract, and several glands.
Conclusion
People and diseases fight to stay alive and learn from each other so they can grow.
The host–pathogen relationship is dynamic. Studies show that human aquaporins are vital in host–pathogen interactions.
This article explains the role of human aquaporins in infectious and non-infectious disorders. Aquaporins in humans let physiologically important substrates into cells, which may change how cells work and, in the long run, how cells fight off disease or fight disease.
Human aquaporins maintain cellular and tissue homeostasis during inflammation, a disease-causing process. Dysregulation, malfunctioning, and malfunction of aquaporins throughout disease development have interesting medicinal uses.
Jump to
Structural Properties Of Human Aquaporins
Human Aquaporins Have Tissue-Specific Distribution And Physiological Relevance
Human Aquaporin Functional Diversity And Regulation
Infectious Diseases - Aquaporin Roles In Infectious Diseases
Involvement Of Aquaporins In Non-Infectious Diseases
Aquaporins As Potential Drug And Diagnostic Targets
People Also Ask
Conclusion

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles