Tandem Heart - What Is It And When Do You Need It?
The Tandem Heart is a percutaneously implanted ventricular assist device (VAD) that offers left atrial to femoral artery bypass at up to 5L/min flow rates. The Tandem Heart employs a transseptal cannula to enable direct unloading of the left heart at blood flow rates adequate to keep patients alive. Furthermore, the Tandem Heart may be utilized for a limited time for high-risk coronary procedures.
Author:Suleman ShahReviewer:Han JuNov 14, 202266 Shares998 Views
Cardiogenic shock in-hospital mortality remains high, ranging from 45 to 60%.
The Tandem Heartis a percutaneously implanted ventricular assist device (VAD) that offers left atrial to femoral artery bypass at up to 5L/min flow rates.
A ventricular assist device (VAD) is an electromechanical device used to support cardiac circulation and replace the function of a failing heart, either partly or totally.
A VAD differs from an artificial cardiac pacemaker in that it pumps blood, while a pacemaker sends electrical impulses to the heart muscle.
Some VADs are intended for short-term use, often for patients recuperating from a myocardial infarction (heart attack) or cardiac surgery; othersare intended for long-term use (months to years to indefinite usage), typically for patients suffering from severe heart failure.
What Is A Tandem Heart?
The Tandem Heart percutaneous ventricular assist device may help patients in cardiogenic shock (until cardiac recovery or as a bridge to definitive treatment) or for temporary use during high-risk coronary operations.
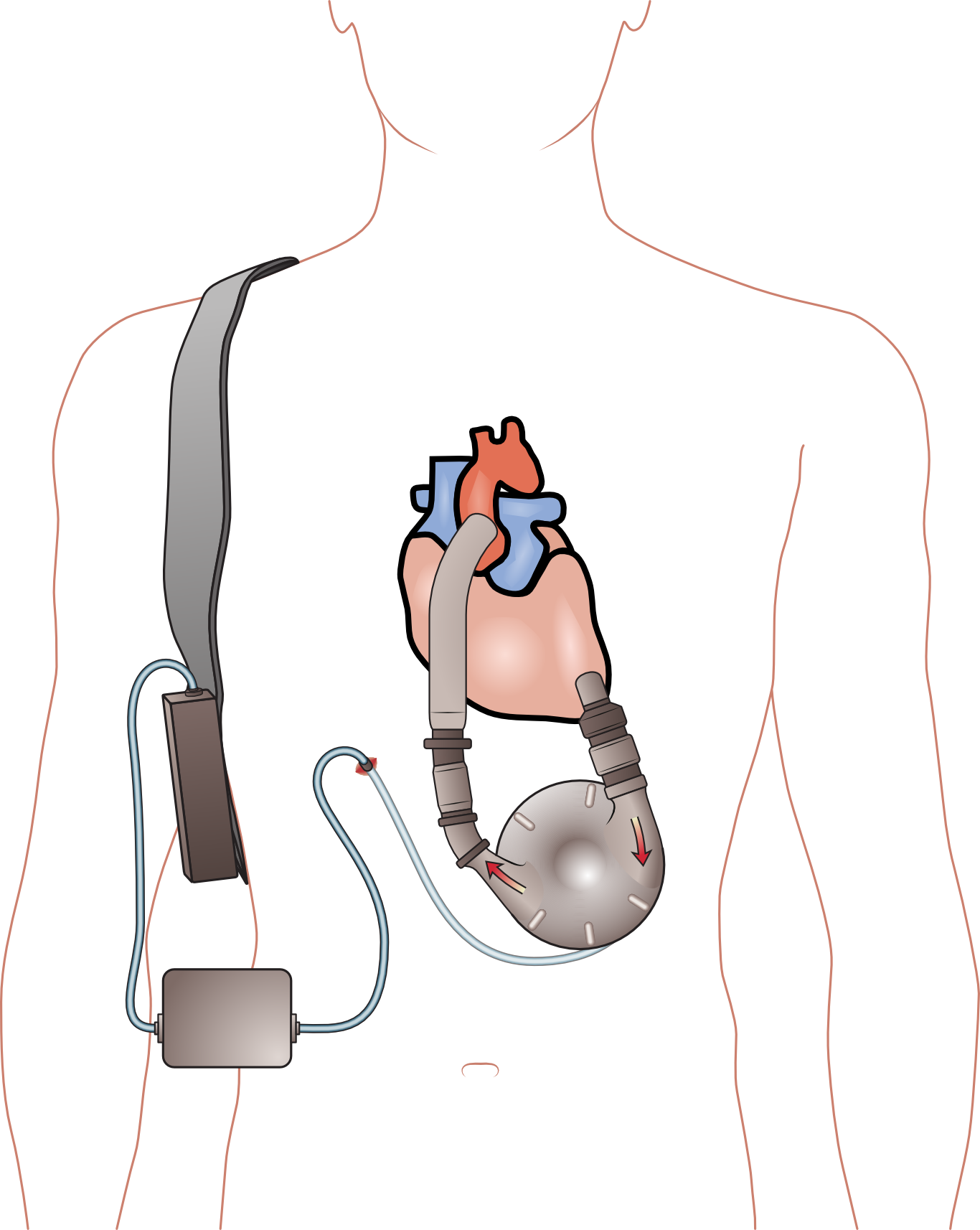
Tandem Heart is a left atrial to femoral artery bypass device that includes a transseptal cannula, arterial cannulae, and a centrifugal blood pump.
The pump has a maximum speed of 7500 rpm and can provide flow rates of up to 4.0 L/min.
The Tandem Heart employs a transseptal cannula to enable direct unloading of the left heart at blood flow rates adequate to keep patients alive.
The technology, which may be installed in the cardiac catheterization laboratory, can help patients until they recover from their cardiac arrest or until another therapeutic option is decided.
Furthermore, the Tandem Heart may be utilized for a limited time for high-risk coronary procedures.

TandemHeart Animation
Ventricular Assist Device (VAD)
In patients with severe heart failure, a ventricular assist device (VAD) is a mechanical device that offers Mechanical Circulatory Support (MCS).
Compared to pharmaceutical treatment alone, VAD therapyimproved outcomes and survival in heart failure patients.
These devices are often utilized as a Bridge-to-Transplant (BTT) in heart transplant eligible patients or as Destination Therapy (DT) in heart transplant-ineligible patients.
VADs are approved for either short-term use in patients recuperating from heart attacks or heart surgery or long-term use (months to years, and in some instances for life) in patients with end-stage heart failure.
SigOver the years, significant progress has been made in manufacturing downsized VADs with better durability, dependability, and lower noise emissions.
Newer (the third-generation use Continuous Flow (CF) pumps, which have lesfewerfficulties than previous (first generation) VADs, which were massive and used pulsatile-flow pumps.
VADs are classified as Left Ventricular Assist Devices (LVADs), Right Ventricular Assist Devices (RVAVADsor BiVentricular Assist Devices (BVADs) de, pending on the heart chamber that requires MCS (BiVADs).
Some VADs are percutaneous or transcutaneous (Impella, TandemHeart), while others (HeartMate II, HeartMate III) are implanted and enable patients to be total.
Ongoing VAD development research focuses on reducing the requirement for a driveline, improving surgical methods, and developing ways for telemetric monitoring of device operation and remote battery charging.
Tandem Heart Procedure
The TandemHeart is a left atrial-to-femoral artery bypass system that consists of a transseptal cannula, arterial cannulae, and a centrifugal blood pump.
All of these components are connected by a transseptal cannula.
A continuous-flow centrifugal pump, the TandemHeart® percutaneous ventricular assist device (pVADTM) (CardiacAssist, Inc.; Pittsburgh, Pa) (made by CardiacAssist, Inc.), can provide flows of up to 4 liters per minute.
The device uses an inflow cannula. It is placed into the left atrium after being introduced into the common femoral vein, traversing the atrial septum, and entering the left atrium.
The oxygenated blood from the left atrium is returned via a cannula that has been implanted percutaneously in the common femoral artery.
This allows the flow to avoid the left ventricle entirely.
As a support device during high-risk percutaneous coronary procedures, the device has been employed in patients who have had a myocardial infarction and are suffering from left ventricular failure. 1,2 In this article, we describe the use of a surgically implanted
TandemHeart is part of a pre-emptive plan for postcardiotomy support in a patient with severe mitral regurgitation, moderate tricuspid regurgitation, and biventricular failure.
The TandemHeart was placed in the patient through cardiac surgery.
Tandem Heart Indications
Prolonged ischemia during the percutaneous coronary intervention (PCI) may have severe hemodynamic effects in individuals with poor left ventricular function and high-risk coronary lesions.
Tandemheart is a short-term circulatory support percutaneous left ventricular assist device.
The Tandem-heart has 9-17 F arterial cannulae and a unique 21 F transseptal cannula and centrifugal blood pump.
The pump takes oxygenated blood from the left atrium and distributes up to 4 liters/min to the arterial circulation at 7500 rpm.
People Also Ask
Can You Do CPR On A Patient With A LVAD?
The use of chest compression in LVAD patients is still debatable.
It is typically not recommended due to the possibility of LVAD dislodgement or regurgitation (from the aorta to the left ventricle).
In one case report, prolonged CPR damaged the percutaneously implanted aortic valve.
Is Tandemheart An ECMO?
The Tandem Heart catheter system may provide unique and improved ECMO delivery alternatives to various populations of patients suffering from cardiopulmonary insufficiency.
What Happens When LVAD Is Turned Off?
When an LVAD is switched off, the patient usually dies within minutes.
However, if the patient has an intrinsic cardiac function, they may survive for many days.
Patients and their families should anticipate these consequences.
To assure the patient's comfort, medications such as opioids and benzodiazepines are administered.
Conclusion
The TandemHeart device is simple to implant and offers a way to either restore heart function or get ongoing help from an implanted left ventricular assist device.

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles