Traumatic Spinal Cord Injury – Injury Mechanism, Diagnosis And Recovery
Traumatic spinal cord injury (SCI) is a life-altering neurological illness with significant socioeconomic consequences for patients and caregivers.
Author:Suleman ShahReviewer:Han JuJul 09, 202269 Shares915 Views
Traumatic spinal cord injuryis a life-altering neurological illness with significant socioeconomic consequences for patients and caregivers. Recent breakthroughs in the medical care of traumatic spinal cord injury have considerably improved the diagnosis, stability, survival rate, and patient well-being.
However, therapyalternatives have been enhanced to improve the neurological outcomes of traumatic spinal cord injury patients. The intricacy of traumatic spinal cord injury pathophysiology and the numerous biochemical and physiological changes in the damaged spinal cord are primarily responsible for this gradual improvement.
As a result, researchers in recent decades have made significant efforts to understand the pathophysiology of traumatic spinal cord injury and unravel the underlying cellular and molecular processes of tissue degeneration and healing in the damaged spinal cord. Various preclinical animal and injury models have been created to mimic better the initial and subsequent damage processes of traumatic spinal cord injury.
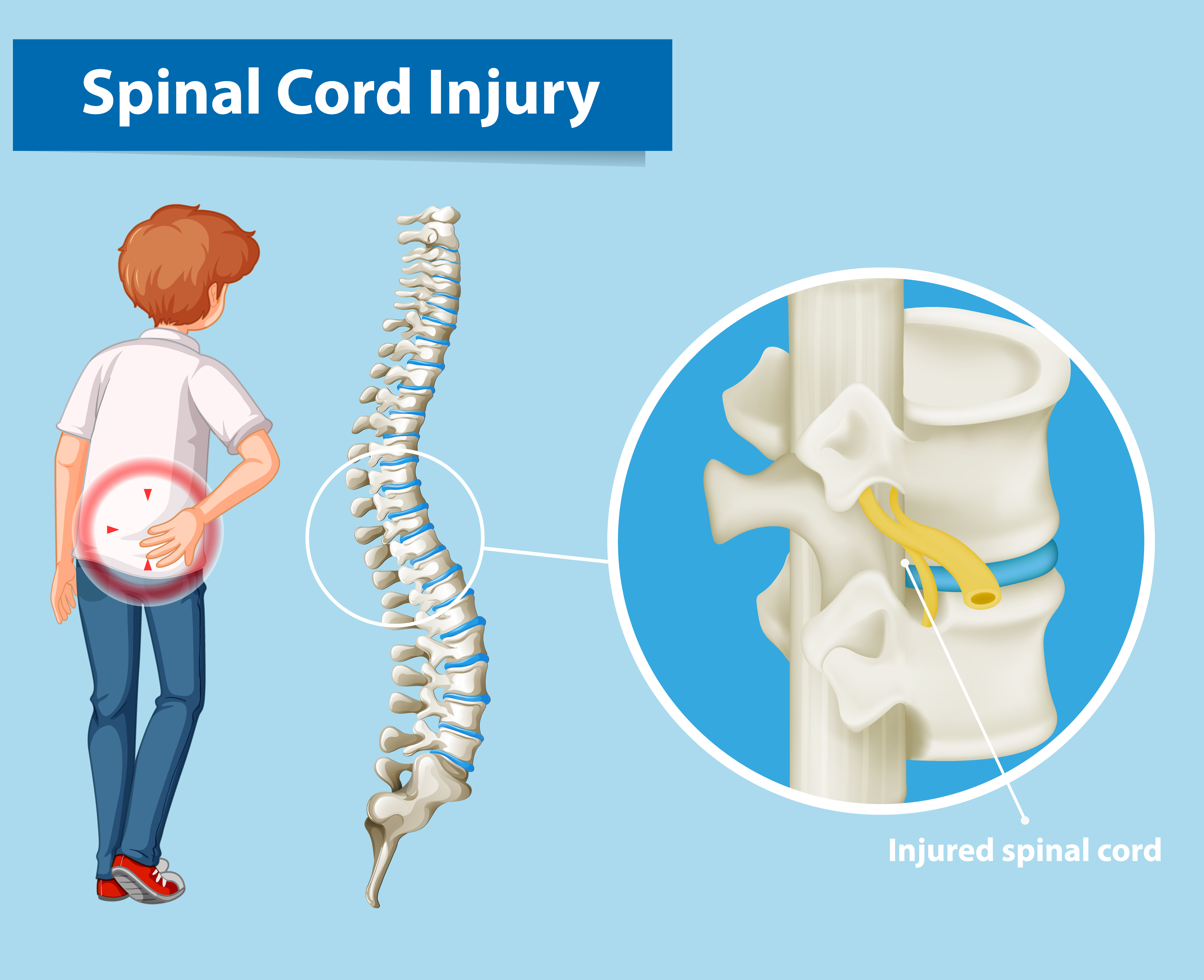
What is a spinal cord injury?
Status Of Traumatic Spinal Cord Injury Cases
12,500 new spinal cord injury cases are reported annually in North America. More than 90% of spinal cord injury instances are traumatic and result from traffic accidents, violence, sports, or falls. Spinal cord injury has a documented male-to-female ratio of 2:1, and it occurs more commonly in adults than children. The age distribution is bimodal, with young people at the top and individuals over 60 at the bottom. spinal cord injury most often affects the cervical level of the spinal cord (50%) and is the most frequent level affected.
Spinal cord injury patients typically survive these catastrophic injuries and live for decades after the original damage because of modern medical techniques and patient care breakthroughs. According to reports on the clinical outcomes of patients who had spinal cord injury in Australia between 1955 and 2006, the survival rates for individuals suffering from tetraplegia and paraplegia are 91.2 and 95.9%, respectively. Individuals with tetraplegia and paraplegia had a 40-year survival rate of 47 and 62%, respectively.
Other areas of injury include the thoracic (35%) and lumbar regions (11%). The amount of damage and retained functions significantly impact the life expectancy of spinal cord injury patients. For example, patients with ASIA Impairment Scale grade D who use a wheelchair for everyday activities have an estimated 75% of typical life expectancy.
Primary Injury Of Spinal Cord
An abrupt, severe impact on the spine fractures or dislocates vertebrae, causing spinal cord injury. Primary damage occurs when "displaced bone fragments, disc materials, and ligaments bruise or shred spinal cord tissue." The most frequent primary injury is impact plus sustained compression, which may result via burst fractures or fracture-dislocation injuries.
In hyperextension injuries, impact with transitory compression is rare. Distraction injuries occur when two neighboring vertebrae are forced apart, stretching and tearing the spinal column. Lacerations and transactions may be caused by missile injuries, severe dislocations, or sharp bone fragment dislocations, ranging from moderate to the total transaction. Military and civilian spinal cord injury results varied.
Battlefield spinal cord injury is frequently caused by blast damage, which affects several spinal cord segments. Blast spinal cord injury has greater severity ratings and lengthier hospital stays.
A study of U.S. military soldiers who experienced spinal cord injury in a conflict zone from 2001 to 2009 found greater severity and worse neurological recovery. Combat injuries cause greater lower lumbar burst fractures and lumbosacral dissociation.
These pressures directly damage ascending and descending spinal cord pathways and disrupt blood vessels and cell membranes, producing spinal shock, systemic hypotension, vasospasm, ischemia, ionic imbalance, and neurotransmitter buildup.
Early spinal cord decompression (24 h post-injury) is the most effective therapeutic therapy to reduce tissue damage after the initial injury. The severity of spinal cord injury depends on the underlying damage.
Spinal Cord Injury Classification System
Functional categorization of spinal cord injury establishes consistent scoring methods to quantify, compare, and link spinal cord injury severity with clinical outcomes. Complete spinal cord injury is rare. In full spinal cord injury, motor and sensory functions are lost below the damage. Clinical categorization of spinal cord injury-related neurological impairments uses many scoring systems. Frankel and colleagues created "Frankel Grade" in 1969.
American Spinal Injury Association (ASIA) Scoring System
ASIA is the most extensively used clinical spinal cord injury rating system. The American Spinal Cord Injury Association created ASIA in 1984 and has revised it. Sensory function is rated 0–2, and motor function 0–5. AIS spans from total loss of sensation and movement to full neurological function. ASIA identifies the degree of neurological damage initially. Except for the top cervical vertebrae, the spinal cord segments and their matching vertebrae are not anatomically aligned throughout the adult spinal cord.
Each vertebra overlaps a spinal cord segment one or two below at thoracic and lumbar levels. A T11 vertebral burst fracture causes neurological deficits at and below the L1 spinal cord segment. NLI is "the most caudal neural level at which sensory and motor functions are normal." If the damage is complete (AIS=A), the "zone of partial preservation" (ZPP) is calculated. ZPP is all segments below NLI with maintained sensory or motor function.
A detailed record of ZPP allows examiners to separate spontaneous from treatment-induced functional improvement, which is critical for assessing therapy success. AIS B involves complete motor and sensory loss below the neurological level of damage (20). If motor function below the lesion is somewhat spared, the AIS score might be C or D. The AIS is graded D when most muscle groups below the damage are strong. ASIA incorporates motor, sensory, and sacral function tests, solving earlier score systems' inadequacies. Validity, repeatability, and accuracy in predicting patient outcomes make ASIA the most acknowledged and trustworthy spinal cord injury clinical assessment system.
Spinal Cord Injury Experimental Models
In the rat, a cystic cavity develops in the spinal cord's core, surrounded by intact white matter. In both rat and human contusive injuries, high-resolution MRI evaluations revealed that spinal cord injury-induced neuroanatomical alterations such as spinal cord atrophy and lesion size were substantially linked with electrophysiological and functional outcomes.
Mice have histopathology that differs from human spinal cord injury in that the lesion site is packed with thick fibrous connective-like tissue. There is a variation in the timing of neutrophil and T cell infiltration in the rat and mouse models of spinal cord injury. Mice have been utilized frequently in spinal cord injury research, regardless of their pathophysiological significance.
In recent years, there has been an increase in interest in using non-human primates and other more giant animals as intermediate pre-clinical models. Large animals are similar to humans in size, neuroanatomy, and physiology. Their size makes them more suitable for medication discovery, bioengineering innovations, electrophysiology, and rehabilitative research.
Transection Models
A complete transaction spinal cord injury model is reasonably simple to replicate. However, it is less applicable to human spinal cord injury. Transection models are ideal for researching axonal regrowth and designing biomaterial scaffolds to bridge the gap between a severed spinal cord's proximal and distal segments. Because these models result in less severe harm and greater amplitude of spontaneous healing, they are less helpful in developing and assessing innovative therapeutics.
Contusive Models
Gruner pioneered the impactor paradigm at New York University in 1992. The original New York University impactor had a metal rod of a set weight (10 g) that could be dropped from a precise height on the exposed spinal cord to cause spinal cord injury. Multicenter Animal Spinal Cord Injury Study III, the most current version, was released in 2012 and incorporated both electromagnetic control and digital recording of impact characteristics. In rats, spinal cord injury has been effectively induced using Multicenter Animal Spinal Cord Injury Study, Infinite Horizon, and Ohio State University impactor devices. Multicenter Animal Spinal Cord Injury Study is a computer-controlled electromagnetic impactor developed in 1987 and modified in 1992 to increase dependability.
The Infinite Horizon impactors may give mild, moderate, or severe spinal cord injury (for example, 100, 150, and 200 kdyn). The inconsistency of these impactors' clamps in retaining the spinal column securely during the impact might produce inconsistent parenchymal damage and neurological impairments.
Compressive Models
In rats and mice, clip compression is the most often utilized compression model of spinal cord injury. A customized aneurism clip with a calibrated closure force is applied to the spinal cord for a defined period in this model (usually 1 min). By altering the power of the clip and the time of compression, the intensity of harm may be calibrated and changed. For example, a 50 g clip for 1 minute usually results in a severe spinal cord injury. Calibrated forceps compression has also been used to instill spinal cord injury in animals.
Calibrated forceps with a spacer is employed in this model to compress the spinal cord bilaterally. This model lacks the initial impact and contusive damage seen in most real traumatic spinal cord injury cases. All compression models, in general, share the identical drawback in that the velocity and quantity of force are unmeasurable.
Spinal Injury Animation
Spinal Cord Injury Mechanism
The secondary injury occurs within minutes of the initial primary injury and may last weeks or months, causing increasing damage to the spinal cord tissue around the lesion site. During research on spinal cord injury in dogs, it was discovered that removing the post-traumatic hematomyelia improved neurological prognosis.
The presence of "biochemical factors" in the necrotic hemorrhagic lesion promotes more spinal cord injury. Secondary injury is still used in the profession to describe various cellular, molecular, and biochemical events that continue to self-destruct spinal cord tissue and hamper neurological recovery after spinal cord injury.
Secondary injury is classified into three time periods: acute, subacute, and chronic. Following spinal cord injury, the acute phase comprises vascular damage, ionic imbalance, neurotransmitter buildup (excitotoxicity), free radical production, calcium influx, lipid peroxidation, inflammation, edema, and necrotic cell death. As the damage continues, apoptosis, demyelination of surviving axons, Wallerian degeneration, axonal dieback, matrix remodeling, and the creation of a glial scar surrounding the injury site occur.
Further changes occur in the chronic damage phase, such as creating a cystic cavity, increasing axonal die-back, and glial scar maturation. The following acute secondary damage components contribute to the pathophysiology of spinal cord injury.
- Ischemia and Hypoxia: One of the early outcomes of initial damage is disruption of spinal cord arterial supply and hypo-perfusion. Within the first several hours after injury, blood flow near the lesion site decreases gradually in rat and monkey models of spinal cord injury. Multiple pathways, including oxygen deprivation, excitotoxicity, ionic imbalance, free radical generation, and necrotic cell death, eventually contribute to cell death and tissue degradation after vascular injury, bleeding, and ischemia.
- Ionic Imbalance, Excitotoxicity, and Oxidative Damage: In the central nervous system, glutamate is the primary excitatory neurotransmitter (CNS). Glutamate binds to ionotropic (NMDA, AMPA, and Kainate receptors) and metabotropic receptors, causing calcium influx inside the cells. Astrocytesmay also release excess glutamate extracellularly when their intracellular Ca2+ levels rise. Mitochondria are crucial in calcium-dependent neuronal death. NMDA receptor overactivity causes mitochondrial calcium overflow in neurons during glutamate-induced excitotoxicity. The mitochondrial calcium uniporter transports Ca2+ into the mitochondria (MCU). Ca2+ excess stimulates a slew of protein kinases and phospholipases shortly after spinal cord injury. Riluzole, a Na+ channel blocker, reduces tissue damage and enhances functional recovery in spinal cord injury, highlighting sodium's central role in secondary injury processes. Increased Na+ concentration activates the Na+/H+ exchanger, increasing intracellular H+. One of the primary pathways of secondary damage after spinal cord injury is lipid and protein oxidation. Lipid and protein oxidation after spinal cord injury has a variety of negative biological implications. These include mitochondrial respiratory and metabolic failure and DNA changes that result in cell death.
- Cell Death: After spinal cord injury, cell death is a significant event in the secondary damage processes that impact neurons and glia. The two primary cell death processes were first recognized as necrosis and apoptosis. A recent study has revealed new types of cell death, such as necroptosis and autophagy. Receptors govern the process of necroptosis. It is activated downstream of the TNF receptor 1 (TNFR1) and relies on RIPK1 and RIPK3. Apoptosis occurs as early as 4 hours after damage in rats and peaks at 7 days. Apoptosis is primarily caused by injury-induced Ca2+ influx, which activates caspases and calpain, enzymes involved in protein degradation. Human post-mortem research demonstrated that Fas-mediated cell death involves oligodendrocyte apoptosis and the inflammatory response after spinal cord injury. Autophagy dysregulation leads to neuronal death and inhibiting it has been linked to neurodegenerative illnesses such as Parkinson's and Alzheimer's. Autophagy helps cells survive by removing harmful proteins and damaged mitochondria.
- Immune Response:Neuroinflammation is a local and systemic secondary damage process. Inflammation was supposed to worsen spinal cord injury. Depending on the timing and immune cell activation, inflammation may be helpful or harmful after spinal cord injury. Injury produces microglia/macrophages/microglia activationand autoantibodies against spinal cord antigens. Astrocytes attract neutrophils via IL-1R1-Myd88. Astrocytes generate TGF and IL-10, which induce a pro-regenerative M2-like phenotype in microglia/macrophages. Within hours after damage, blood neutrophils invade the spinal cord. Their removal hinders healing and rehabilitation. Neutrophil depletion alters cytokines, chemokines, and growth factors. Within 2–3 days post-spinal cord injury, microglia/macrophages colonize the damaged spinal cord. Macrophages come from blood monocytes, which are bone marrow myeloid offspring. T and B cells are critical to spinal cord injury immunological recovery. Autoreactive T cells kill neurons and glial cells. Pro-inflammatory cytokine and chemokine production by T lymphocytes affect brain cell activity and survival. Myelin-specific proteins like myelin basic protein stimulate circulating T lymphocytes in spinal cord injury and Multiple sclerosispatients. Spinal cord injury suppresses B-cell activation and antibody generation. Immunomodulatory Breg B cells help heal the spinal cord. Autoreactive immune cells worsen post-spinal cord injury cardiovascular, renal, and reproductive dysfunctions.
- Glial Scar Tissue:Glial scarring is a multifactorial phenomenon caused by various populations in the damaged spinal cord. Both resident and invading inflammatory cells have a role in glial activation and scar formation. Fibroblasts, pericytes, and ependymal cells, in addition to glial and immunological cells, contribute to its construction. Activated astrocytes are key players in the creation of the glial scar. Reactive astrogliosis is also required for blood-brain-barrier rebuilding. By inhibiting this mechanism, leukocyte infiltration, cell death, myelin degradation, and functional recovery are all hampered. Chondroitin sulfate proteoglycans (CSPGs) are well recognized for their contribution to the glial scar's inhibitory function in axonal regeneration. Following injury, CSPGs are strongly upregulated and reach a peak of expression two weeks later. The inhibitory effects of the astrocytic glial scar on axonal regeneration have lately been called into question after spinal cord injury. Using multiple transgenic mice models, Sofroniew's and colleagues discovered that spontaneous axon regeneration did not occur after scar ablation or prevention.
People Also Ask
What Causes A Traumatic Spinal Cord Injury?
Road traffic accidents fall, and violence is the primary cause of spinal cord damage (including attempted suicide). Work or sports-related injuries account for a significant fraction of traumatic spinal cord damage.
What Are The Two Types Of Spinal Cord Injuries?
The majority of cases may be categorized into two forms of spinal cord damage: full spinal cord injury and incomplete spinal cord injury. A complete spinal cord injury causes permanent damage to the damaged portion of the spinal cord. Complete spinal cord damage results in paraplegia or tetraplegia.
What Are The Three Types Of Spinal Cord Injuries?
There are three types of spinal cord injuries: tetraplegia, paraplegia, and triplegia.
What Is The Most Serious Permanent Effect Of Spinal Cord Trauma?
A spinal cord injury is one of the most devastating ailments a person may have. This form of physical damage may cause paraplegia, or the inability to move the lower body, or quadriplegia, or the inability to move the body at all.
Final Words
Traumatic spinal cord injury has diverse and complicated pathogenesis. While pre-clinical research on spinal cord injury has continued for over a century, our knowledge of spinal cord injury processes has grown significantly in the last few decades. This is mainly owing to the development of novel transgenic and preclinical animal models, which have aided in the quick discovery of spinal cord injury mechanisms. Although spinal cord injury research has made significant progress, more effort has to be made to transfer findings from animal studies to therapeutic applications in people.

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles