Osteosarcoma - How Can Animal Models Help Us Study It?
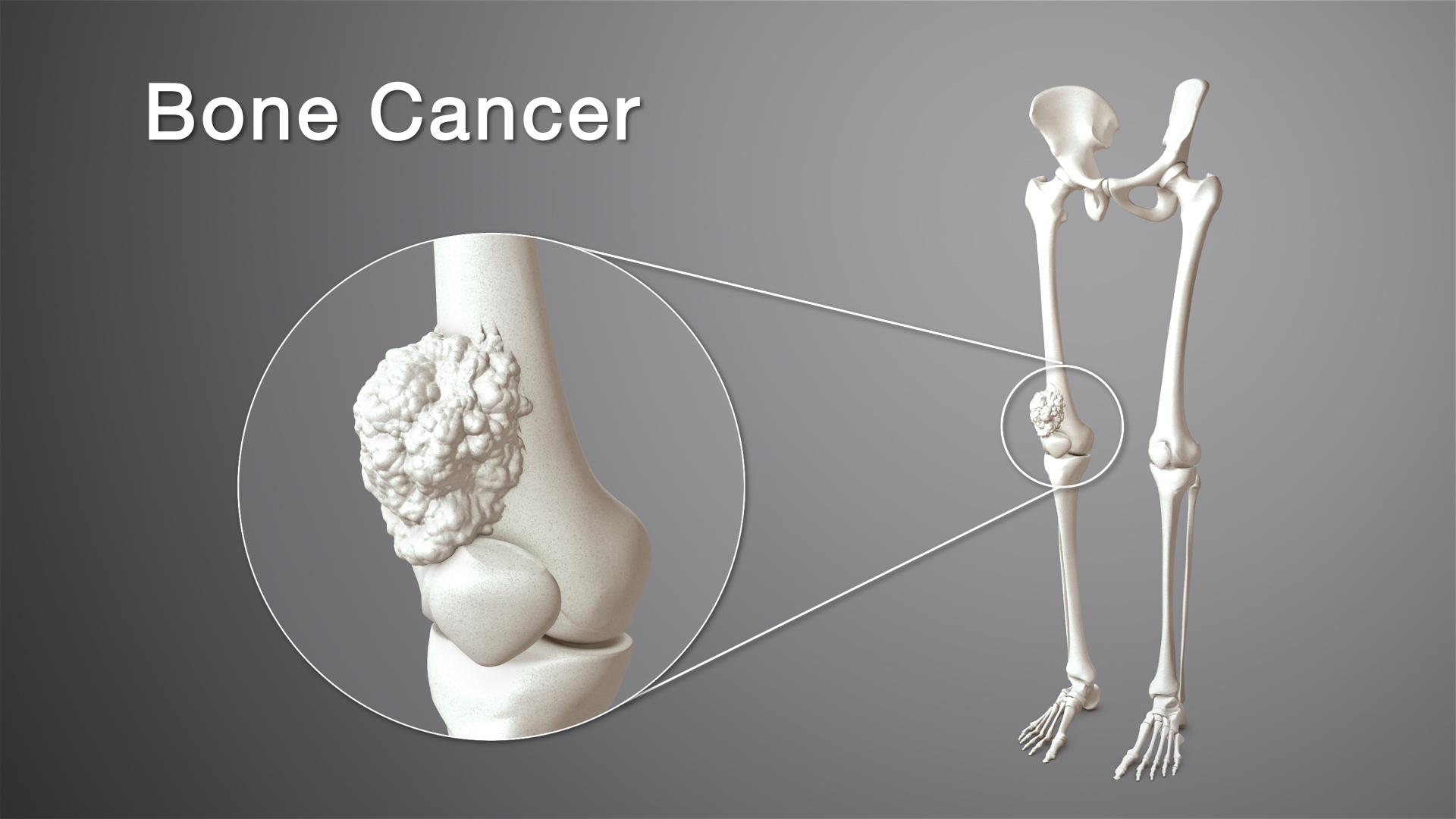
Osteosarcoma, a type of cancer that causes bones to grow abnormally, has a good outlook. Osteosarcoma is the most common bone cancer, but it only makes up less than 1% of all cancer cases in the United States. After the first 10 years of life, when bones are still growing, there is a second peak in the number of cases in people who are over 60.
Author:Suleman ShahReviewer:Han JuJul 07, 20220 Shares120 Views
Osteosarcoma, a type of cancer that causes bones to grow abnormally, has a good outlook.
Osteosarcoma is the most common bone cancer, but it only makes up less than 1% of all cancer cases in the United States.
After the first 10 years of life, when bones are still growing, there is a second peak in the number of cases in people who are over 60.
90% of osteosarcoma in children and teens starts in the intramedullary space of long bones in the lower limbs, usually in the metaphyses.
The data show that this has something to do with active growth plates. Osteosarcoma usually comes back in older people after a short time of getting better in people ages 25 to 59. It is usually caused by Paget's disease or radiation exposure.
For example, this could mean that the real cause of the problem is different for younger and older patients.
In general, there are three main subtypes of osteosarcoma: osteoblastic, fibroblastic, and chondroblastic. The most common type is osteoblastic, then fibroblastic, and then chondroblastic.
What Are The Symptoms Of Osteosarcoma?
Osteosarcoma's symptoms and indicators vary depending on the location of the tumor. Pain in the bones, bone features, limping, swelling, joints that are less mobile are some of the most prevalent symptoms.
It is possible to experience bone pain in a variety of different ways. A dull soreness or pain may keep your youngster up at night. If your child exhibits any of the symptoms listed above, including bone discomfort, you should have them get their muscles checked. When a person is diagnosed with osteosarcoma, their muscles in the affected limb may appear smaller than those in the unaffected limb.
Osteosarcoma-like aches in the legs might be caused by normal bone development. In contrast, the difficulties of adolescence typically subside around the age of 13 or 14. Doctors should be consulted if your child's bone discomfort or swelling lasts longer than their first growth spurt or if the pain is causing them significant harm.
Osteosarcoma or Bone Cancer - Yale Medicine Explains
Etiology Of Osteosarcoma
Because of its complex karyotype and lack of recurrent translocations, osteosarcoma is difficult to classify genetically. Researchers have discovered many genes that may have a role in the disease's development and dissemination. It's difficult to interpret these findings because of the large range of chromosomal alterations in the osteosarcoma genome.
Genetic changes in osteosarcoma are random, however those with Li-Fraumeni and retinoblastic syndromes are more prone to get the cancer. P53 has somatic deletions or point mutations in half of all human osteosarcomas, and half of these mutations result in the loss of the remaining allele.
RB allele mutations are found in over seventy percent of osteosarcomas. More cancers contain RB point mutations than tumors with RB homozygous deletions. Also, a number of changes have been discovered that interfere with the RB pathway. There are 22 percent of osteosarcomas with either an INK4a/ARF locus loss of function or an increase in CDK4 gene expression. According to these modifications, it is likely that the cell cycle checkpoints in osteosarcoma are out of control on a regular basis.
More representative models are required to learn more about osteosarcoma biology. One reason for this is due to a tumor's aggressiveness and early beginning, which are all characteristics of osteosarcomas.
Animal Models In Osteosarcoma
Osteosarcoma has a complex karyotype and is free of recurrent translocations. Researchers have discovered a number of genes that may have a role in the disease's development and spread through genomic analysis. Osteosarcoma's genome has a wide range of chromosomal alterations, making it difficult to interpret these findings. Most genetic changes in osteosarcoma are random, but those with the Li-Fraumeni and retinoblastoma syndromes are more likely to have the disease. On average, P53 is altered in half of all osteosarcoma patients, leading to allele loss for those patients who remain genetically unaffected.
Additionally, the RB allele is altered in about seventy percent of osteosarcomas. Only 6% of cancers contain point mutations in RB, but 23.3% of tumors have homozygous deletions of RB. Also, a number of alterations to the RB pathway have been discovered. ' The INK4a/ARF locus or CDK4 gene amplification is found in 22% of osteosarcomas, for example. Osteosarcoma appears to have a problem with the G1/S and G2/M checkpoints in the cell cycle, based on the frequency of these changes.
Osteosarcoma biology can only be better understood through the use of more diverse animal models. To put it another way, osteosarcoma is a cancer that has no known cure due to its aggressiveness and unpredictable origins.
Canine Models
Unlike people, large dogs are more likely to develop spontaneous osteosarcoma. This makes the dog an ideal model for studying human disease. In termsof histology and gene expression, canine osteosarcoma is identical to human tumors. Age at onset and disease prevalence stand out as the most significant distinctions between the two scenarios.
It is estimated that about 10,000 instances of osteosarcoma in dogs occur each year in the United States. This disease usually affects older, large breed dogs (6–12 years old). After surgery alone, the median disease-free interval is four months; after surgery and chemotherapy, it is 13 months. For this reason, it is possible to mimic metastasis growth and progression in a relatively short amount of time, as well as test new treatment strategies. Human osteosarcoma pathogenesis genes such as P53, RB, and PTEN appear to be implicated in canine osteosarcoma pathogenesis.
In spite of the fact that the human and canine models of osteosarcoma share many similarities, there are several caveats. One important difference between humans and canines is that osteosarcoma is more common in older dogs than in adolescents. Another factor to consider is the fact that some breeds of dogs have distinct germ-line mutations in certain genes that may influence the development and response to osteosarcoma treatment.
Osteosarcoma
Secondary Osteosarcoma After Radiation
Exposure of rats and mice to chemical and radioactive carcinogens sparked the development of rodent osteosarcoma models. There were a number of notable findings, among them the high prevalence of osteosarcoma in rats treated with P32-orthophosphate. As a result of these models, researchers have developed tumors that mirror human cancer and cell lines that can be used to supplement human osteosarcoma research. Because most osteosarcomas in people are sporadic, a therapy-induced disease, the carcinogen-induced murine model provides a better representation of the human disease despite its great penetrance.
Xenotransplantation Studies
The development and use of xenograft and allograft models of human and murine osteosarcoma cells implanted into immunocompromised mice is well documented in the scientific literature.
Within a few days or weeks of implantation, the injected cells develop into a solid tumor. Because of its quick start, low cost, and ease of use and maintenance, these systems are already a common tool in oncological research. To explore primary and secondary tumor progression, donor-derived cells from osteosarcoma may spread to the lungs. The approach's primary drawback is the use of fully formed osteosarcoma cells, which leaves open the question of how the tumor began and its genesis.
As a result, interactions between the tumor and its microenvironment may be lost if the disease is established in a recipient animal through direct introduction. As long as the implanted cell line is not metastatic, it is impossible to analyze how the disease spreads in rodents.
There are still many groups that have used this model successfully to uncover the parameters that influence osteosarcoma migration and more significantly to screen medicines with tumoricidal potential. The high rate of occurrence and reproducibility of the subcutaneous cell suspension injection model allow for accurate titration of cell numbers in the inoculum to measure the tumorigenic potential of the injected cells. This model has distinct advantages.
Genetically Engineered Mouse Models
Osteosarcoma is one of the most well-studied sarcomas, as evidenced by the development of various mice models for the study of this condition. Gene targeting technologies have made it possible to modify the expression of individual genes (either by deleting or enhancing their function) in the mouse.
The P53 and RB mutations in hereditary and sporadic human osteosarcoma have been replicated in a number of mouse osteosarcoma models. When P53 was deleted from the germ line, it resulted in an incidence of osteosarcoma of 4% among homozygotes and 25% among heterozygotes, demonstrating the relevance of P53 in driving osteosarcoma.
The early fatality in the homozygous null population is most likely to blame for this surprising tumor development ratio. This is due to the quick development of other cancers (mainly lymphomas), the long latency of osteosarcoma, and the need to sacrifice the mice before osteosarcoma onset, which in many situations limits the usefulness of these models. Using P53 knock-in mice with a mutant copy of P53R172H (equivalent to the R175H hot-spot mutation in humans) as a model for tumor development and metastasis to other organs, researchers were able to further demonstrate the importance of P53. Rb homologous deletion is embryonic fatal and heterozygotes are not susceptible to osteosarcoma, in contrast to mice with germ-line deletions of Rb developing osteosarcoma.
Our ability to construct specialized models of mesenchymal osteogenic lineage has been considerably increased by the use of conditional gene regulation and the availability of tissue specific Cre expressing animal lines. Penetrant osteosarcoma models have been generated via the loss of P53, either alone or in combination with the Rb pathway disruption.
Targeted Therapies In Osteosarcoma
Numerous mice models have been developed for osteosarcoma, making it one of the better-studied sarcomas despite its unique karyotype. There has been an increase in the number of gene targeting technologies that allow scientists to control the expression of individual genes.
The P53 and RB alterations in hereditary and sporadic human osteosarcoma have been replicated in a variety of mouse osteosarcoma models. Osteosarcoma incidence was 4 percent in homozygous P53 null mice and 25 percent in heterozygous P53 animals, demonstrating the role of mutated P53 in promoting osteosarcoma development. But this surprising ratio of tumor growth is attributable to the early fatality seen in the homozygous null population, which is likely to be the cause. This is due to the rapid development of other cancers (mainly lymphomas), the longer latency of osteosarcoma, and the need to sacrifice the mice before osteosarcoma onset, which in many cases limits the usefulness of these models. When P53-knock-in mice with the R175H hot-spot mutation (which occurs in human cancers) form primary tumors as well as spread to other organs, tumor examination reveals the critical involvement of P53. Germ-line deletion of Rb prevented osteosarcoma in mice, however, because homologous deletion of Rb is embryonic fatal, heterozygotes are not at risk for developing osteosarcoma.
Our ability to construct particular models of mesenchymal osteogenic lineage that more closely match human osteosarcoma has been considerably increased by the use of conditional gene regulation and the availability of tissue specific Cre expressing mouse lines. Osteosarcoma models based on the loss of P53, with or without the disruption of the Rb pathway, have been generated in most cases.
People Also Ask
What Is The Main Cause Of Osteosarcoma?
As far as we know, the specific cause of osteosarcoma remains a mystery; nonetheless, it is widely accepted that the disease is caused by genetic abnormalities in bone cells.
Is Osteosarcoma Cancer Curable?
Osteosarcoma can now be cured in roughly 3 out of 4 patients if it hasn't migrated to other parts of the body. Almost everyone who has limb-sparing surgery recovers the ability to use their arm or leg. Osteosarcoma patients often require physical rehabilitation for months after their operation.
Who Is Most Likely To Get Osteosarcoma?
Osteosarcoma is most common in children and young adults between the ages of 10 and 30, particularly during the period of rapid growth that occurs during this time. This shows that tumor genesis may be linked to fast bone expansion. In middle age, the risk decreases, but it climbs again in the elderly (usually over the age of 60).
Where Does Osteosarcoma Usually Start?
An osteosarcoma is a bone cancer that starts in the cells that create bones. It is a type of osteosarcoma. Osteosarcoma most commonly affects the long bones, such as the limbs, but it can also occur in the arms or hands. Soft tissue outside of the bone is affected in just a small percentage of cases.
Conclusion
The lack of predisposition conditions or precursor lesions in osteosarcoma biology makes it difficult for researchers to understand the disease. When it comes to studying the fundamental mechanisms that drive tumor development, progression, metastatic occurrences, and therapeutic interventions, human tissue is limited.
Animal models fill this void. In spite of the lack of complete realism in these models, the study of osteosarcoma animal models has provided insights into tumor initiation and progression, as well as the cellular and molecular profiles of tumors. Genetic mutations that accelerate tumor initiation (P53), as well as co-operative genetic mutations that increase illness incidence, have been discovered using gene knockout experiments (RB, c-FOS).

Suleman Shah
Author
Suleman Shah is a researcher and freelance writer. As a researcher, he has worked with MNS University of Agriculture, Multan (Pakistan) and Texas A & M University (USA). He regularly writes science articles and blogs for science news website immersse.com and open access publishers OA Publishing London and Scientific Times. He loves to keep himself updated on scientific developments and convert these developments into everyday language to update the readers about the developments in the scientific era. His primary research focus is Plant sciences, and he contributed to this field by publishing his research in scientific journals and presenting his work at many Conferences.
Shah graduated from the University of Agriculture Faisalabad (Pakistan) and started his professional carrier with Jaffer Agro Services and later with the Agriculture Department of the Government of Pakistan. His research interest compelled and attracted him to proceed with his carrier in Plant sciences research. So, he started his Ph.D. in Soil Science at MNS University of Agriculture Multan (Pakistan). Later, he started working as a visiting scholar with Texas A&M University (USA).
Shah’s experience with big Open Excess publishers like Springers, Frontiers, MDPI, etc., testified to his belief in Open Access as a barrier-removing mechanism between researchers and the readers of their research. Shah believes that Open Access is revolutionizing the publication process and benefitting research in all fields.

Han Ju
Reviewer
Hello! I'm Han Ju, the heart behind World Wide Journals. My life is a unique tapestry woven from the threads of news, spirituality, and science, enriched by melodies from my guitar. Raised amidst tales of the ancient and the arcane, I developed a keen eye for the stories that truly matter. Through my work, I seek to bridge the seen with the unseen, marrying the rigor of science with the depth of spirituality.
Each article at World Wide Journals is a piece of this ongoing quest, blending analysis with personal reflection. Whether exploring quantum frontiers or strumming chords under the stars, my aim is to inspire and provoke thought, inviting you into a world where every discovery is a note in the grand symphony of existence.
Welcome aboard this journey of insight and exploration, where curiosity leads and music guides.
Latest Articles
Popular Articles